Difference between revisions of "Main Page"
| (42 intermediate revisions by 2 users not shown) | |||
| Line 2: | Line 2: | ||
{| class="MainPageBG" style="margin: auto; width: 95%; border-spacing:0px;" | {| class="MainPageBG" style="margin: auto; width: 95%; border-spacing:0px;" | ||
|- | |- | ||
| − | | style="width: | + | | style="width:55%;" |<center><span style="font-size:175%; line-height: 0.2em; vertical-align:top;"><big><span style="color:#008566">Welcome to '''ENVIRO'''</span> <span style="color:#762a87">'''Wiki'''</span></big></span><br /><br /><br /><span style="font-size:150%; color:#008566; line-height: 0.2em; vertical-align:top;"> Peer Reviewed. Accessible. Written By Experts</span></center> |
| − | | style="width: | + | | style="width:40%;" |<center><span style="font-size:110%; vertical-align:top;"> ''Developed and brought to you by '' <br>[[File:MainLogo-serdp-estcp.png|link=https://www.serdp-estcp.org |frameless|center|350px]]</span>''<span style="font-size:140%; vertical-align:top;">Your Environmental Information Gateway</span>'' |
</center> | </center> | ||
|- | |- | ||
| − | |<span style="line-height: 0.3em;"> The goal of ENVIRO | + | |<span style="width:55%; line-height: 0.3em;"> The goal of ENVIRO Wiki is to make scientific and engineering research results more accessible to environmental professionals, facilitating the permitting, design and implementation of environmental projects. Articles are written and edited by invited experts (see [[Contributors]]) to summarize current knowledge for the target audience on an array of topics, with cross-linked references to reports and technical literature. </span> |
|<center><span style="font-size:130%"><br />[[#Table of Contents|See Table of Contents]]</span> | |<center><span style="font-size:130%"><br />[[#Table of Contents|See Table of Contents]]</span> | ||
</center> | </center> | ||
| Line 17: | Line 17: | ||
{| role="presentation" id="mp-upper" style="margin: auto; width: 95%; margin-top:3px; border-spacing: 0px; " | {| role="presentation" id="mp-upper" style="margin: auto; width: 95%; margin-top:3px; border-spacing: 0px; " | ||
<!-- TODAY'S FEATURED ARTICLE --> | <!-- TODAY'S FEATURED ARTICLE --> | ||
| − | | id="mp-left" class="MainPageBG" style="width: | + | | id="mp-left" class="MainPageBG" style="width:55%; padding:0; vertical-align:top; color:#000;" | |
| − | <h2 id="mp-tfa-h2" style="margin:0.5em; background:#cef2e0; font-family:inherit; font-size:120%; font-weight:bold; border:1px solid #a3bfb1; color:#000; padding:0.2em 0.4em;"> Featured article: | + | <h2 id="mp-tfa-h2" style="margin:0.5em; background:#cef2e0; font-family:inherit; font-size:120%; font-weight:bold; border:1px solid #a3bfb1; color:#000; padding:0.2em 0.4em;"> Featured article: Photoactivated Reductive Defluorination - PFAS Destruction</h2> |
| − | <div id="mp-tfa" style="padding:0.0em 1.0em;">[[File: | + | <div id="mp-tfa" style="padding:0.0em 1.0em;">[[File:WittFig1.png|550px|left|link=Photoactivated Reductive Defluorination - PFAS Destruction]]<dailyfeaturedpage></dailyfeaturedpage> |
| − | [[ | + | [[Photoactivated Reductive Defluorination - PFAS Destruction|(Full article...)]] </div> |
| style="border:1px solid transparent;" | | | style="border:1px solid transparent;" | | ||
| Line 31: | Line 31: | ||
<slideshow sequence="random" transition="fade" refresh="7500"> | <slideshow sequence="random" transition="fade" refresh="7500"> | ||
| − | [[File:WH Picture1.JPG|thumb|center|x350px|link= | + | [[File:WH Picture1.JPG|thumb|center|x350px|link=Matrix Diffusion|Molecular diffusion slowly transports solutes into clay-rich, lower permeability zones]] |
[[File:WH Picture2.JPG|thumb|center|x350px|link=Subgrade Biogeochemical Reactor (SBGR)|Typical subgrade biogeochemical reactor (SBGR) layout. The SBGR is an in situ remediation technology for treatment of contaminated source areas and groundwater plume hot spots<br/>]] | [[File:WH Picture2.JPG|thumb|center|x350px|link=Subgrade Biogeochemical Reactor (SBGR)|Typical subgrade biogeochemical reactor (SBGR) layout. The SBGR is an in situ remediation technology for treatment of contaminated source areas and groundwater plume hot spots<br/>]] | ||
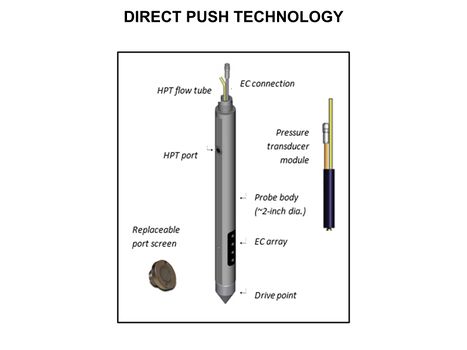
[[File:WH Picture3.JPG|thumb|center|x350px|link=Direct Push Logging|An Hydraulic Profiling Tool (HPT) log with electrical conductivity (EC) on left, injection pressure in middle, and flow rate on the right]] | [[File:WH Picture3.JPG|thumb|center|x350px|link=Direct Push Logging|An Hydraulic Profiling Tool (HPT) log with electrical conductivity (EC) on left, injection pressure in middle, and flow rate on the right]] | ||
| Line 52: | Line 52: | ||
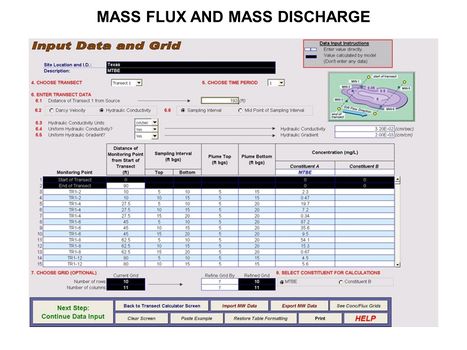
[[File:WH Picture20.JPG|thumb|center|x350px|link=Mass_Flux_and_Mass_Discharge|Data input screen for ESTCP Mass Flux Toolkit]] | [[File:WH Picture20.JPG|thumb|center|x350px|link=Mass_Flux_and_Mass_Discharge|Data input screen for ESTCP Mass Flux Toolkit]] | ||
[[File:WH Picture21.JPG|thumb|center|x350px|link=Bioremediation_-_Anaerobic_Design_Considerations|Amendment addition for biobarrier]] | [[File:WH Picture21.JPG|thumb|center|x350px|link=Bioremediation_-_Anaerobic_Design_Considerations|Amendment addition for biobarrier]] | ||
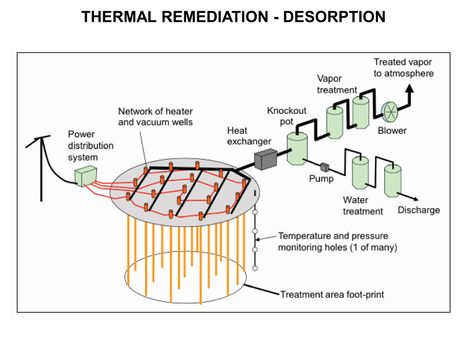
| − | [[File:WH Picture22.JPG|thumb|center|x350px|link= | + | [[File:WH Picture22.JPG|thumb|center|x350px|link=Thermal Conduction Heating (TCH)|Thermal Remediation - Desorption schematic]] |
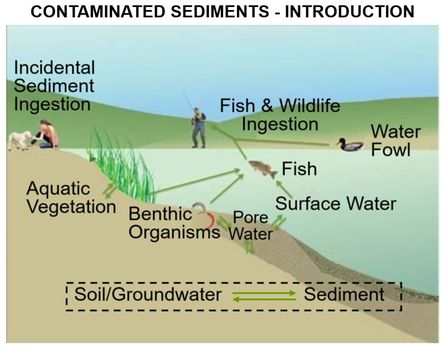
[[File:WH_Picture23.jpg|thumb|center|x350px|link=Contaminated_Sediments_-_Introduction |Key exposure pathways for human health risk from contaminated sediments]] | [[File:WH_Picture23.jpg|thumb|center|x350px|link=Contaminated_Sediments_-_Introduction |Key exposure pathways for human health risk from contaminated sediments]] | ||
[[File:WH_Picture24.jpg|thumb|center|x350px|link=Perfluoroalkyl_and_Polyfluoroalkyl_Substances_(PFAS)| The PFAS family of compounds]] | [[File:WH_Picture24.jpg|thumb|center|x350px|link=Perfluoroalkyl_and_Polyfluoroalkyl_Substances_(PFAS)| The PFAS family of compounds]] | ||
| Line 79: | Line 79: | ||
*[[Vapor Intrusion (VI)]] | *[[Vapor Intrusion (VI)]] | ||
**[[Vapor Intrusion - Separation Distances from Petroleum Sources]] | **[[Vapor Intrusion - Separation Distances from Petroleum Sources]] | ||
| − | **[[Vapor Intrusion – Sewers and Utility Tunnels as Preferential Pathways]] | + | **[[Vapor Intrusion – Sewers and Utility Tunnels as Preferential Pathways|Vapor Intrusion - Sewers and Utility Tunnels as Preferential Pathways]] |
<u>'''[[Characterization, Assessment & Monitoring]]'''</u> | <u>'''[[Characterization, Assessment & Monitoring]]'''</u> | ||
| Line 86: | Line 86: | ||
*[[Compound Specific Isotope Analysis (CSIA)|Compound Specific Isotope Analysis (CSIA)]] | *[[Compound Specific Isotope Analysis (CSIA)|Compound Specific Isotope Analysis (CSIA)]] | ||
*[[Direct Push (DP) Technology]] | *[[Direct Push (DP) Technology]] | ||
| − | **[[Direct Push Logging | | + | **[[Direct Push Logging | Direct Push Logging]] |
| − | **[[Direct Push Sampling | | + | **[[Direct Push Sampling | Direct Push Sampling]] |
*[[Geophysical Methods | Geophysical Methods]] | *[[Geophysical Methods | Geophysical Methods]] | ||
**[[Geophysical Methods - Case Studies | Case Studies]] | **[[Geophysical Methods - Case Studies | Case Studies]] | ||
| Line 96: | Line 96: | ||
*[[Molecular Biological Tools - MBTs | Molecular Biological Tools (MBTs)]] | *[[Molecular Biological Tools - MBTs | Molecular Biological Tools (MBTs)]] | ||
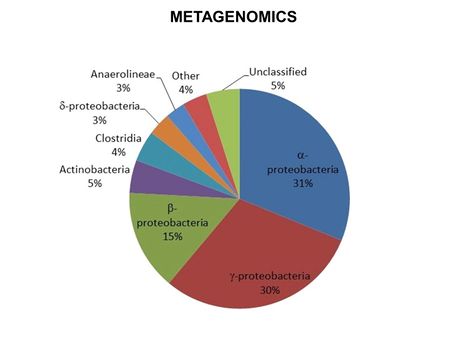
**[[Metagenomics]] | **[[Metagenomics]] | ||
| + | **[[Proteomics and Proteogenomics]] | ||
**[[Quantitative Polymerase Chain Reaction (qPCR)]] | **[[Quantitative Polymerase Chain Reaction (qPCR)]] | ||
**[[Stable Isotope Probing (SIP)]] | **[[Stable Isotope Probing (SIP)]] | ||
*[[Natural Attenuation in Source Zone and Groundwater Plume - Bemidji Crude Oil Spill | Natural Attenuation in Source Zone and Groundwater Plume -<br /> Bemidji Crude Oil Spill]] | *[[Natural Attenuation in Source Zone and Groundwater Plume - Bemidji Crude Oil Spill | Natural Attenuation in Source Zone and Groundwater Plume -<br /> Bemidji Crude Oil Spill]] | ||
| − | <u>'''[[Climate Change Primer]]'''</u> | + | <u>'''[[Climate Change Primer | Climate Change]]'''</u> |
| + | *[[Climate Change Effects on Wildlife]] | ||
*[[Downscaled High Resolution Datasets for Climate Change Projections]] | *[[Downscaled High Resolution Datasets for Climate Change Projections]] | ||
| + | *[[Infrastructure Resilience]] | ||
| + | *[[Predicting Species Responses to Climate Change with Population Models]] | ||
| + | *[[Restoration of Ecological Function in Terrestrial Systems Impacted by Invasive Species]] | ||
<u>'''[[Coastal and Estuarine Ecology]]'''</u> | <u>'''[[Coastal and Estuarine Ecology]]'''</u> | ||
| Line 109: | Line 114: | ||
| style="width:33%; vertical-align:top; " | | | style="width:33%; vertical-align:top; " | | ||
| − | |||
<u>'''[[Contaminated Sediments - Introduction | Contaminated Sediments]]'''</u> | <u>'''[[Contaminated Sediments - Introduction | Contaminated Sediments]]'''</u> | ||
| + | *[[Contaminated Sediment Risk Assessment]] | ||
*[[In Situ Treatment of Contaminated Sediments with Activated Carbon]] | *[[In Situ Treatment of Contaminated Sediments with Activated Carbon]] | ||
*[[Mercury in Sediments]] | *[[Mercury in Sediments]] | ||
*[[Passive Sampling of Sediments]] | *[[Passive Sampling of Sediments]] | ||
| + | *[[Sediment Capping]] | ||
<u>'''[[Light Non-Aqueous Phase Liquids (LNAPLs)]]'''</u> | <u>'''[[Light Non-Aqueous Phase Liquids (LNAPLs)]]'''</u> | ||
| Line 124: | Line 130: | ||
<u>'''[[Munitions Constituents]]'''</u> | <u>'''[[Munitions Constituents]]'''</u> | ||
| + | *[[Munitions Constituents - Abiotic Reduction| Abiotic Reduction]] | ||
*[[Munitions Constituents - Alkaline Degradation| Alkaline Degradation]] | *[[Munitions Constituents - Alkaline Degradation| Alkaline Degradation]] | ||
*[[Munitions Constituents - Composting| Composting]] | *[[Munitions Constituents - Composting| Composting]] | ||
*[[Munitions Constituents - Deposition | Deposition]] | *[[Munitions Constituents - Deposition | Deposition]] | ||
*[[Munitions Constituents - Dissolution | Dissolution]] | *[[Munitions Constituents - Dissolution | Dissolution]] | ||
| + | *[[Metal(loid)s - Small Arms Ranges]] | ||
*[[Passive Sampling of Munitions Constituents| Passive Sampling]] | *[[Passive Sampling of Munitions Constituents| Passive Sampling]] | ||
| − | *[[ | + | *[[Munitions Constituents – Photolysis | Photolysis]] |
*[[Munitions Constituents - Soil Sampling | Soil Sampling]] | *[[Munitions Constituents - Soil Sampling | Soil Sampling]] | ||
*[[Munitions Constituents - Sorption | Sorption]] | *[[Munitions Constituents - Sorption | Sorption]] | ||
| Line 138: | Line 146: | ||
*[[Monitored Natural Attenuation (MNA) of Chlorinated Solvents| MNA of Chlorinated Solvents]] | *[[Monitored Natural Attenuation (MNA) of Chlorinated Solvents| MNA of Chlorinated Solvents]] | ||
| + | *[[Monitored Natural Attenuation (MNA) of Fuels| MNA of Fuels]] | ||
*[[Monitored Natural Attenuation (MNA) of Metal and Metalloids| MNA of Metals and Metalloids]] | *[[Monitored Natural Attenuation (MNA) of Metal and Metalloids| MNA of Metals and Metalloids]] | ||
| − | |||
*[[Natural Source Zone Depletion (NSZD)]] | *[[Natural Source Zone Depletion (NSZD)]] | ||
*[[Monitored Natural Attenuation - Transitioning from Active Remedies| Transitioning from Active Remedies]] | *[[Monitored Natural Attenuation - Transitioning from Active Remedies| Transitioning from Active Remedies]] | ||
<u>'''[[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS)]]'''</u> | <u>'''[[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS)]]'''</u> | ||
| + | |||
*[[PFAS Ex Situ Water Treatment]] | *[[PFAS Ex Situ Water Treatment]] | ||
*[[PFAS Soil Remediation Technologies]] | *[[PFAS Soil Remediation Technologies]] | ||
*[[PFAS Sources]] | *[[PFAS Sources]] | ||
*[[PFAS Transport and Fate]] | *[[PFAS Transport and Fate]] | ||
| + | *[[PFAS Treatment by Electrical Discharge Plasma]] | ||
| + | *[[Photoactivated Reductive Defluorination - PFAS Destruction|Photoactivated Reductive Defluorination]] | ||
<u>'''[[Regulatory Issues and Site Management]]'''</u> | <u>'''[[Regulatory Issues and Site Management]]'''</u> | ||
| Line 191: | Line 202: | ||
<u>'''[[Soil & Groundwater Contaminants]]'''</u> | <u>'''[[Soil & Groundwater Contaminants]]'''</u> | ||
| + | *[[1,2,3-Trichloropropane]] | ||
*[[1,4-Dioxane]] | *[[1,4-Dioxane]] | ||
*[[Chlorinated Solvents]] | *[[Chlorinated Solvents]] | ||
| Line 198: | Line 210: | ||
*[[Petroleum Hydrocarbons (PHCs)]] | *[[Petroleum Hydrocarbons (PHCs)]] | ||
*[[Polycyclic Aromatic Hydrocarbons (PAHs)]] | *[[Polycyclic Aromatic Hydrocarbons (PAHs)]] | ||
| − | |||
|} | |} | ||
|} | |} | ||
|} | |} | ||
Revision as of 20:50, 27 June 2024
Peer Reviewed. Accessible. Written By Experts |
Your Environmental Information Gateway |
| The goal of ENVIRO Wiki is to make scientific and engineering research results more accessible to environmental professionals, facilitating the permitting, design and implementation of environmental projects. Articles are written and edited by invited experts (see Contributors) to summarize current knowledge for the target audience on an array of topics, with cross-linked references to reports and technical literature. | See Table of Contents |
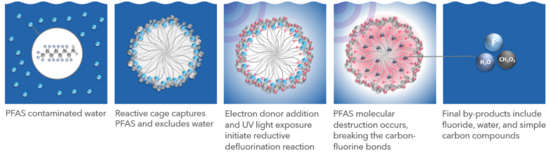
Featured article: Photoactivated Reductive Defluorination - PFAS DestructionPhotoactivated Reductive Defluorination (PRD) is a PFAS destruction technology predicated on ultraviolet (UV) light-activated photochemical reactions. The destruction efficiency of this process is enhanced by the use of a surfactant to confine PFAS molecules in self-assembled micelles. The photochemical reaction produces hydrated electrons from an electron donor that associates with the micelle. These highly reactive hydrated electrons have the energy required to cleave fluorine-carbon and other molecular bonds resulting in the final products of fluoride, water, and simple carbon molecules. Since the reaction is performed at ambient temperature and pressure, there are limited concerns regarding environmental health and safety or volatilization of PFAS compared to heated and pressurized systems. Due to the reductive nature of the reaction, there is no formation of unwanted byproducts resulting from oxidative processes. The PRD reaction rate decreases in water matrices with high levels of total dissolved solids (TDS). The PRD reaction rate decreases in water matrices with very low UV transmissivity. Low UV transmissivity (i.e., < 1 %) prevents the penetration of UV light into the solution, such that the utilization efficiency of UV light decreases. Due to the first-order kinetics of PRD, destruction of PFAS is generally most energy efficient when paired with pre-concentration technologies, such as foam fractionation (FF), nanofiltration, reverse osmosis, or resin/carbon adsorption, that remove PFAS from water.
(Full article...)
|
Enviro Wiki Highlights |