|
|
| Line 1: |
Line 1: |
| − | ==Supercritical Water Oxidation (SCWO)== | + | ==Estimating PCE/TCE Abiotic First-Order Reductive Dechlorination Rate Constants in Clayey Soils Under Anoxic Conditions== |
| − | Supercritical water oxidation (SCWO) is a single step [[Wikipedia: Wet oxidation | wet oxidation]] process that transforms organic matter into water, carbon dioxide and, depending on the waste undergoing treatment, an inert mineral solid residue. The process is highly effective and can treat a variety of wet wastes without dewatering. The SCWO technology allows for the complete destruction of persistent and toxic organic contaminants such as [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) | perfluoroalkyl and polyfluoroalkyl substances (PFAS)]], [[1,4-Dioxane | 1,4-dioxane]], and many more.
| + | The U.S. Department of Defense (DoD) faces many challenges in restoring aquifers at contaminated sites, often due to back-diffusion of tetrachloroethene (PCE) and trichloroethene (TCE) from low-permeability clay zones. The uptake, storage, and subsequent long-term release of these dissolved contaminants from clays are key processes in understanding the longevity, intensity, and risks associated with many persistent chlorinated ethene groundwater plumes. Although naturally occurring abiotic and biotic dechlorination processes in clays may reduce stored contaminant mass and significantly aid natural attenuation, no standardized field method currently exists to verify or quantify these reactions. It is critical to remediation design efforts to demonstrate and validate a cost-effective in situ approach for assessing these dechlorination processes using first-order rate constants. An approach was developed and applied across eight DoD sites to support Remedial Project Managers (RPMs) and regulators in evaluating natural attenuation potential in clay-rich environments. |
| | <div style="float:right;margin:0 0 2em 2em;">__TOC__</div> | | <div style="float:right;margin:0 0 2em 2em;">__TOC__</div> |
| | | | |
| | '''Related Article(s):''' | | '''Related Article(s):''' |
| | | | |
| − | * [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS)]] | + | *[[Monitored Natural Attenuation (MNA)]] |
| − | * [[PFAS Transport and Fate]] | + | *[[Monitored Natural Attenuation (MNA) of Chlorinated Solvents]] |
| − | * [[Chlorinated Solvents]] | + | *[[Monitored Natural Attenuation - Transitioning from Active Remedies]] |
| | + | *[[Matrix Diffusion]] |
| | + | *[[REMChlor - MD]] |
| | | | |
| − | '''Contributor(s):''' [[Kobe Nagar]] and [[Dr. Marc Deshusses]] | + | '''Contributors:''' Dani Tran, Dr. Charles Schaefer, Dr. Charles Werth |
| | | | |
| − | '''Key Resource(s):''' | + | '''Key Resource:''' |
| − | | + | *Schaefer, C.E, Tran, D., Nguyen, D., Latta, D.E., Werth, C.J., 2025. Evaluating Mineral and In Situ Indicators of Abiotic Dechlorination in Clayey Soils<ref name="SchaeferEtAl2025"/> |
| − | *Treatment of municipal sewage sludge in supercritical water: A review<ref name="Qian2016">Qian, L., Wang, S., Xu, D., Guo, Y., Tang, X., and Wang, L., 2016. Treatment of municipal sewage sludge in supercritical water: A review. Water Research, 89, pp. 118-131. [https://doi.org/10.1016/j.watres.2015.11.047 DOI: 10.1016/j.watres.2015.11.047] Free download from: [https://www.researchgate.net/profile/Shuzhong-Wang/publication/284563832_Treatment_of_Municipal_Sewage_Sludge_in_Supercritical_Water_a_Review/links/5d9b63b6299bf1c363fef63e/Treatment-of-Municipal-Sewage-Sludge-in-Supercritical-Water-a-Review.pdf ResearchGate]</ref>. | |
| − | | |
| − | *Supercritical Water Oxidation – Current Status of Full-scale Commercial Activity for Waste Destruction<ref name="Marrone2013">Marrone, P.A., 2013. Supercritical Water Oxidation – Current Status of Full-scale Commercial Activity for Waste Destruction. Journal of Supercritical Fluids, 79, pp. 283-288. [https://doi.org/10.1016/j.supflu.2012.12.020 DOI: 10.1016/j.supflu.2012.12.020] Author’s manuscript available from: [https://semspub.epa.gov/work/06/9545963.pdf US EPA]</ref>.
| |
| | | | |
| | ==Introduction== | | ==Introduction== |
| − | Supercritical water oxidation (SCWO) is an [[Wikipedia: Advanced oxidation process | advanced oxidation process]] that holds enormous potential for the treatment of a wide range of organic wastes, in particular concentrated wet wastes in slurries such as biosolids, sludges, agricultural wastes, chemical wastes with recalcitrant chemicals such as [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS)| perfluoroalkyl and polyfluoroalkyl substances (PFAS)]], and many more. SCWO relies on the unique reactivity and transport properties that occur when an aqueous waste stream is brought above the critical point of water (374°C and 218 atm, or 704°F and 3200 psi, see phase diagram in Figure 1). [[Wikipedia: Supercritical fluid | Supercritical water]] is a dense single phase with transport properties similar to those of a gas, and solvent properties comparable to those of a non-polar solvent<ref name="Tassaing2002">Tassaing, T., Danten, Y., and Besnard, M., 2002. Infrared spectroscopic study of hydrogen bonding in water at high temperature and pressure, Journal of Molecular Liquids, 101(1-3), pp. 149-158. [https://doi.org/10.1016/S0167-7322(02)00089-2 DOI: 10.1016/S0167-7322(02)00089-2]</ref>. Oxygen is fully soluble in supercritical water, resulting in extremely rapid and complete oxidation of all organics to carbon dioxide, clean water (that can be reused), and some non-leachable inorganic salts.
| + | Cost-effective methods are needed to verify the occurrence of natural dechlorination processes and quantify their dechlorination rates in clays under ambient in situ conditions in order to reliably predict their long-term influence on plume longevity and mass discharge. However, accurately determining these rates is challenging due to slow reaction kinetics, the transient nature of transformation products, and the interplay of biotic and abiotic mechanisms within the clay matrix or at clay-sand interfaces. Tools capable of quantifying these reactions and assessing their role in mitigating plume persistence would be a significant aid for long-term site management. |
| | | | |
| − | For SCWO to be economical, the heat from the oxidation reaction is recovered and used in part to heat the influent stream, while the excess heat can be converted to electricity. Depending on the concentration of waste in the feedstock, SCWO reactors can be operated autothermally, i.e., no outside input of heat is required. Typical reaction times are in the order of 2-10 seconds, resulting in SCWO systems that are quite compact compared to other technologies (see Table 1). The process does not generate harmful by-products such as nitrogen oxides (NOx) or Sulfur oxides (SOx), carbon monoxide (CO), or odors<ref Name="Bermejo">Bermejo, M.D. and Cocero, M.J., 2006. Supercritical water oxidation: A technical review. AIChE Journal, 52(11) pp. 3933-3951. [https://doi.org/10.1002/aic.10993 DOI: 10.1002/aic.10993]</ref>. Typically, if present, ammonia and organic nitrogen in the waste undergoing treatment are converted to nitrogen gas, while phosphorous precipitates as phosphates and can be recovered. When [[Wikipedia: Halogen | halogen]] containing contaminants are treated (e.g., [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS)| PFAS]]), halogen-carbon bonds are generally broken and [[Wikipedia: Halide | halide]] anions are released in solution (e.g., F- when treating PFAS or Cl- when treating [[Wikipedia: Trichloroethylene | trichloroethene (TCE)]] and [[Wikipedia: Tetrachloroethylene | tetrachloroethene (PCE)]]). | + | For reductive abiotic dechlorination under anoxic conditions, a 1% hydrochloric acid (HCl) extraction of a sample of native clay coupled with X-ray diffraction (XRD) data can be used as a screening level tool to estimate reductive dechlorination rate constants. These rate constants can be inserted into fate and transport models such as [[REMChlor - MD]]<ref>Falta, R., and Wang, W., 2017. A semi-analytical method for simulating matrix diffusion in numerical transport models. Journal of Contaminant Hydrology, 197, pp. 39-49. [https://doi.org/10.1016/j.jconhyd.2016.12.007 doi: 10.1016/j.jconhyd.2016.12.007] [[Media: FaltaWang2017.pdf | Open Access Manuscript]]</ref><ref>Kulkarni, P.R., Adamson, D.T., Popovic, J., Newell, C.J., 2022. Modeling a well-charactized perfluorooctane sulfate (PFOS) source and plume using the REMChlor-MD model to account for matrix diffusion. Journal of Contaminant Hydrology, 247, Article 103986. [https://doi.org/10.1016/j.jconhyd.2022.103986 doi: 10.1016/j.jconhyd.2022.103986] [[Media: KulkarniEtAl2022.pdf | Open Access Manuscript]]</ref> to quantify abiotic dechlorination impacts within clay aquitards on chlorinated solvent plumes. Thus, determination of the abiotic reductive dechlorination rate constant for a particular clayey soil can be readily utilized to provide a more accurate assessment of aquifer cleanup timeframes for groundwater plumes that are being sustained by contaminant back-diffusion. |
| | | | |
| − | ==Advantages and Disadvantages== | + | ==Recommended Approach== |
| − | There are many advantages to SCWO treatment. SCWO is a destructive treatment in that the compounds treated are mineralized to simple elements or harmless molecules (e.g., water and carbon dioxide) rather than just being transferred to another medium. Another advantage is the absence of reaction by-products, incompletely oxidized contaminants or unreacted harmful oxidants (e.g., ozone). SCWO is an extremely rapid and effective reaction (typical reaction times are in the order of 5-10 seconds) making it possible to build systems that are very compact and have a high throughput. SCWO is also a very clean process. The highly oxidizing environment makes it possible to effectively treat all sorts of organic contaminants, often recalcitrant to other processes, with very high (>99%) destruction efficiencies. This includes treatment of trace contaminants, slurries of biosolids, waste oil, food wastes, plastics, or emerging contaminants such as PFAS or 1,4-dioxane. Also, the relatively moderate temperatures (380-600°C) compared to other destructive technologies such as incineration, combined with the presence of supercritical water prevent the formation of NOx and SOx compounds. Lastly, SCWO treatment does not require drying of the waste, and both liquids and slurries can be treated using SCWO.
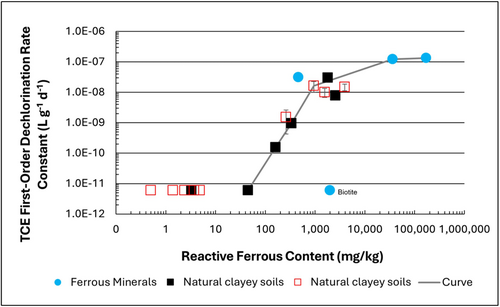
| + | [[File: TranFig1.png | thumb | 500 px | Figure 1: First-order rate constants for abiotic reductive dechlorination of TCE under anaerobic conditions. Circles are data from Schaefer ''et al.'', 2021<ref>Schaefer, C.E., Ho, P., Berns, E., Werth, C., 2021. Abiotic dechlorination in the presence of ferrous minerals. Journal of Contaminant Hydrology, 241, 103839. [https://doi.org/10.1016/j.jconhyd.2021.103839 doi: 10.1016/j.jconhyd.2021.103839] [[Media: SchaeferEtAl2021.pdf | Open Access Manuscript]]</ref>, filled squares from Schaefer ''et al.'', 2018<ref name="SchaeferEtAl2018"/>, and Schaefer ''et al.'', 2017<ref>Schaefer, C.E., Ho., Gurr, C., Berns, E., Werth, C., 2017. Abiotic dechlorination of chlorinated ethenes in natural clayey soils: impacts of mineralogy and temperature. Journal of Contaminant Hydrology, 206, pp. 10-17. [https://doi.org/10.1016/j.jconhyd.2017.09.007 doi: 10.1016/j.jconhyd.2017.09.007] [[Media: SchaeferEtAl2017.pdf | Open Access Manuscript]]</ref>, and open squares from Schaefer ''et al.'', 2025<ref name="SchaeferEtAl2025"/>. ]] |
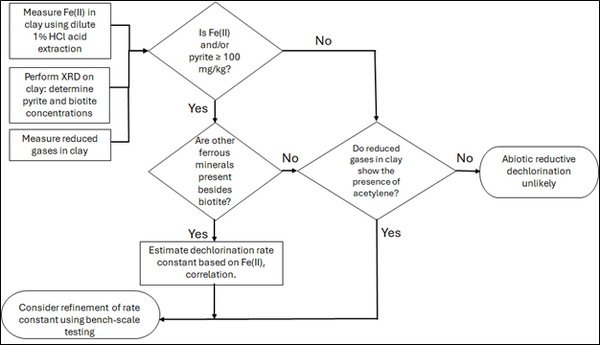
| | + | [[File: TranFig2.png | thumb | 600 px | Figure 2: Flowchart diagram of field screening procedures]] |
| | + | The recommended approach builds upon the methodology and findings of a recent study<ref name="SchaeferEtAl2025">Schaefer, C.E., Tran, D., Nguyen, D., Latta, D.E., Werth, C.J., 2025. Evaluating Mineral and In Situ Indicators of Abiotic Dechlorination in Clayey Soils. Groundwater Monitoring and Remediation, 45(2), pp. 31-39. [https://doi.org/10.1111/gwmr.12709 doi: 10.1111/gwmr.12709]</ref>, emphasizing field-based and analytical techniques to quantify abiotic first-order reductive dechlorination rate constants for PCE and TCE in clayey soils under anoxic conditions. Key components of this evaluation are listed below: |
| | + | #<u>Zone Identification:</u> The focus of the investigation should be to delineate clayey zones adjacent to hydraulically conductive zones. |
| | + | #<u>Ferrous Mineral Quantification:</u> Assess ferrous mineral context in clay via 1% HCl extraction at ambient temperature over a 10-minute interval. |
| | + | #<u>Mineralogical Characterization:</u> Conduct XRD analysis with the specific intent of identifying the presence of pyrite and biotite. |
| | + | #<u>Reduced Gas Analysis:</u> Measurement of reduced gases such as acetylene, ethene, and ethane concentrations in clay samples. Gas-tight sampling devices (e.g., En Core® soil samplers by En Novative Technologies, Inc.) should be used to ensure sample integrity during collection and transport. |
| | | | |
| − | There are several disadvantages to SCWO treatment. First, a significant amount of energy needs to be expended to bring the oxidant and the waste undergoing treatment to the critical point of water. Although a large fraction of this energy can be efficiently recovered in heat exchangers, compensating for heat losses constrains SCWO to the treatment of concentrated wastes with sufficient organic content for the exothermic oxidation reaction to provide the necessary heat. Typically, a minimum calorific content of around 2 MJ/kg (which generally corresponds to a chemical oxygen demand of about 120-150 g/L) is needed for autothermal operation. For more dilute streams, external heating or supplementation of fuel (diesel, alcohol, waste oil, etc.) can be implemented, but it can rapidly become cost prohibitive. Thus, SCWO is currently not economical for very large volumes (>50,000 gallon/day) of very dilute waste streams. A second limitation is related to the pumping of the waste. Because the process is conducted at high pressure (240 bars or 3500 psi), positive displacement pumps are required. This limits SCWO to liquids and slurries that can be pumped. Waste streams that contain excessive grit or abrasive materials, and soils cannot currently be processed using SCWO.
| + | Clay samples should be collected within a few centimeters of the high-permeability interface, with optional additional sampling further inward. For mineralogical analysis, a defined interval may be collected and subsequently subsampled. To preserve sample integrity, exposure to air should be minimized during collection, transport, and handling. Homogenization should occur within an anaerobic chamber, and if subsamples are required for external analysis, they must be shipped in gas-tight, anaerobic containers. |
| | | | |
| − | The many appealing benefits of supercritical water processing have stimulated engineers and entrepreneurs to invest significant efforts and resources in the development of the technology. Today, after roughly 30 years of development, commercial deployment is on the horizon<ref name="Marrone2013"/>. Technical challenges that have slowed down commercial deployment of SCWO are linked to the complex nature of a high-pressure, high-temperature process. Critical issues include reactor materials selection to resist corrosion (typically high nickel alloys are used), reactor designs and construction to withstand the corrosive nature of the reactive mass, dealing with highly exothermic reactions at high pressure and high temperature, plugging of the reactor by minerals deposits, and energy recovery for autothermal operation. Another challenge was the unrealistic goal of some companies entering the SCWO market to produce power from waste streams (often wastewater sludge) at a competitive cost (3-5 cents/kWh). This was not feasible with the available technology, which led to several business failures.
| + | Estimation of the abiotic reductive first-order rate constant for PCE and TCE is based on the “reactive” ferrous content in the clay. Reactive ferrous content (Fe(II)<sub>r</sub>) is estimated as shown in Equation 1: |
| | | | |
| − | The value proposition of treating recalcitrant wastes using SCWO is markedly different, especially in today’s context of increasing liability for trace levels of emerging contaminants such as PFAS. SCWO may prove to be the optimal treatment technology for many highly concentrated aqueous waste streams.
| + | ::'''Equation 1:''' <big>''Fe(II)<sub><small>r</small></sub> = DA + XRD<sub><small>pyr</small></sub> - XRD<sub><small>biotite</small></sub>''</big> |
| | | | |
| − | ==State of the Art==
| + | where ''DA'' is the ferrous content from the dilute acid (1% HCl) extraction, ''XRD<sub><small>pyr</small></sub>'' is the pyrite content from XRD analysis, and ''XRD<sub><small>biotite</small></sub>'' is the biotite content from XRD analysis<ref name="SchaeferEtAl2025"/>. |
| − | Relatively few large scale SCWO systems exist. Researchers at Duke University ([http://sanitation.pratt.duke.edu/community-treatment/about-community-treatment-project Deshusses lab]) have designed and built a prototype pilot-scale SCWO system housed in a standard 20-foot shipping container (Figure 2). This project was funded by the Reinvent the Toilet program of the [https://www.gatesfoundation.org/ Bill and Melinda Gates Foundation]. The pilot system is a continuous process designed to treat 1 ton of sludge per day at 10-20% dry solids content. The unit has been undergoing testing at Duke since early 2015. The design includes moderate preheating of the waste slurry, followed by mixing with supercritical water (~600°C) and air, which serves as the oxidant. This internal mixing rapidly brings the waste undergoing treatment to supercritical conditions thereby minimizing corrosion and the risks of waste charring and associated reactor plugging. The organics in the sludge are rapidly oxidized to CO<sub>2</sub>, while the heat of oxidation is recovered to heat the influent waste. The reactor is a single tubular reactor. The high supercritical fluid velocity in the system helps with controlling mineral salts deposition in the reactor. The system is well instrumented, and operation is controlled using a supervisory control and data acquisition (SCADA) system with historian software for trends analysis and reporting of key performance indicators (e.g., temperatures and pressures, pollutant destruction). Experiments conducted with this pilot plant have shown effective treatment of a wide variety of otherwise problematic wastes such as primary, secondary and digested sludge slurries, landfill leachate (see Figure 3), animal waste, and co-contaminants including waste oil, food wastes, and plastics. The results are consistent with other SCWO studies and show very rapid treatment of all wastes with near complete conversion (often >99.9%) of organics to CO<sub>2</sub>. Total nitrogen and phosphorous removal are generally over 95% and 98%, respectively. Emerging contaminants such as pharmaceuticals, [[Perfluoroakyl and Polyfluoroalkyl Substances (PFAS) |PFAS]], [[1,4-Dioxane | 1,4-dioxane]] and [[Wikipedia: Microplastics | microplastics]] are also treated with destruction generally exceeding 99%.
| |
| | | | |
| − | Early projections for treatment costs (Capital Expenditures + Operating Expenditures) for slurries are in the range of $12 to $90 per ton (or $0.04 to $0.37 per gallon) depending on system scale and contaminant concentration, with a majority of the cost coming from amortizing the equipment. These cost projections make SCWO treatment very competitive compared to other treatment technologies for high-strength wastes. When treating large volumes of water, combining SCWO with another technology (e.g., nanofiltration, reverse osmosis, or adsorption onto GAC) should be considered so that only the concentrated brines or spent sorbent are treated using SCWO, thereby increasing the cost effectiveness of the overall treatment.
| + | Abiotic dechlorination is unlikely to contribute to mitigating contaminant back-diffusion when reactive ferrous iron (Fe(II)<sub><small>r</small></sub>) concentrations are below 100 mg/kg (Figure 1). For Fe(II)<sub><small>r</small></sub> above 100 mg/kg, the first-order rate constant for PCE and TCE reductive dechlorination can be estimated using the correlation shown in Figure 1<ref name="SchaeferEtAl2018">Schaefer, C.E., Ho, P., Berns, E., Werth, C., 2018. Mechanisms for abiotic dechlorination of trichloroethene by ferrous minerals under oxic and anoxic conditions in natural sediments. Environmental Science and Technology, 52(23), pp.13747-13755. [https://doi.org/10.1021/acs.est.8b04108 doi: 10.1021/acs.est.8b04108]</ref><ref>Borden, R.C., Cha, K.Y., 2021. Evaluating the impact of back diffusion on groundwater cleanup time. Journal of Contaminant Hydrology, 243, Article 103889. [https://doi.org/10.1016/j.jconhyd.2021.103889 doi: 10.1016/j.jconhyd.2021] [[Media: BordenCha2021.pdf | Open Access Manuscript]]</ref>. The rate constant exhibits a strong positive correlation with the logarithm of reactive Fe(II) content (Pearson’s ''r'' = 0.82), with a slope of 4.7 × 10⁻⁸ L g⁻¹ d⁻¹ (log mg kg⁻¹)⁻¹. |
| | | | |
| − | ==SCWO for the Treatment of PFAS and AFFF==
| + | Figure 2 presents a decision flowchart designed to evaluate the significance and extent of abiotic reductive dechlorination. By applying Equation 1 to the dilute acid extractable Fe(II) plus measured mineral species data from clay samples, the reactive ferrous iron content (Fe(II)<sub><small>r</small></sub>) can be quantified, enabling a streamlined assessment of the extent to which abiotic processes are contributing to the mitigation of contaminant back-diffusion. |
| − | Several reports have indicated that PFAS can be treated using SCWO<ref name="Kucharzyk2017">Kucharzyk, K.H., Darlington, R., Benotti, M., Deeb, R. and Hawley, E., 2017. Novel treatment technologies for PFAS compounds: A critical review. Journal of Environmental Management, 204(2), pp. 757-764. [https://doi.org/10.1016/j.jenvman.2017.08.016 DOI: 10.1016/j.jenvman.2017.08.016] Manuscript available from: [https://www.researchgate.net/profile/Katarzyna_kate_Kucharzyk/publication/319125507_Novel_treatment_technologies_for_PFAS_compounds_A_critical_review/links/5a06590b4585157013a3be77/Novel-treatment-technologies-for-PFAS-compounds-A-critical-review.pdf ResearchGate]</ref>. Several runs treating biosolids known to contain PFAS as well as dilutions of pure [[Wikipedia: Firefighting foam | aqueous film forming foam (AFFF)]] have also been conducted with the Duke SCWO system. Typical results are shown in Table 2. They indicate very effective treatment performance, with for example 110,000 ng/L PFOS in the feed reduced to 0.79 ng/L in the effluent, and many other PFAS reduced to below their detection limits. No HF was found in the effluent gas, and all the fluorine from the destroyed PFAS was accounted for as fluoride in the effluent water. These results show the ability of the SCWO process to destroy PFAS to levels well below the EPA health advisory levels of 70 ng/L for PFOS and PFOA. The ESTCP project [https://www.serdp-estcp.org/Program-Areas/Environmental-Restoration/ER20-5350/ER20-5350 ER20-5350]<ref name="Deshusses2020">Deshusses, M.A., 2020. Supercritical Water Oxidation (SCWO) for Complete PFAS Destruction. ER20-5350. [https://www.serdp-estcp.org/Program-Areas/Environmental-Restoration/ER20-5350/ER20-5350 Project website]</ref> launched in June 2020 will assess the technical feasibility of using supercritical water oxidation (SCWO) for the complete destruction of PFAS in a variety of relevant waste streams and will evaluate the cost effectiveness of the treatment.
| |
| | | | |
| | + | If Fe(II)r is ≥ 100 mg/kg, a first-order dechlorination rate constant can be estimated and subsequently used within a contaminant fate and transport model. However, if acetylene is detected in the clay, even with Fe(II)r less than 100 mg/kg, then bench-scale testing using methods similar to those described in a recent study<ref name="SchaeferEtAl2025"/> is recommended, as such results would likely be inconsistent with those shown in Figure 1, suggesting some other mechanism might be involved, or that the system mineralogy might be more complex than anticipated. Even if Fe(II)r ≥ 100 mg/kg, confirmatory bench-scale testing may be conducted for additional verification and to refine estimation of the abiotic dechlorination rate constant. |
| | | | |
| | + | ==Summary and Recommendations== |
| | + | The approach outlined above is intended to serve as a generalized guide for practitioners and site managers to cost-effectively determine the extent to which beneficial abiotic reductive dechlorination reactions are likely occurring in low permeability (e.g., clayey) zones. This approach may be contraindicated if co-contaminants are present. It is currently unclear whether other classes of potentially reactive chemicals, such as trinitrotoluene (TNT) or chlorinated ethanes, could interact competitively with PCE and TCE. |
| | | | |
| − | Three technologies are well demonstrated for removal of [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) | PFAS]] from drinking water and non-potable groundwater (as described below):
| + | In addition, it remains unclear how other classes of compounds such as per- and polyfluoroalkyl substances (PFAS) may interact or sorb with ferrous minerals and potentially inhibit abiotic dechlorination reactions. Coupling these recommended activities with conventional site investigation tasks would provide an opportunity to perform many of the up-front screening activities with minimal additional project costs. It is important to note that the guidance proposed herein pertains to particularly low permeability media. Sites with complex or varying lithology, where the mineralogy and/or redox conditions may vary, might require evaluation of multiple samples to provide appropriate site-wide information. |
| − |
| |
| − | * membrane filtration including [[wikipedia: Reverse osmosis | reverse osmosis (RO)]] and [[Wikipedia: Nanofiltration | nanofiltration (NF)]]
| |
| − | * granular [[Wikipedia: Activated carbon | activated carbon]] (GAC) and powdered activated carbon (PAC) adsorption
| |
| − | * [[wikipedia: Ion_exchange | anion exchange (IX)]]
| |
| | | | |
| − | However, these technologies are less demonstrated for removal of PFAS from more complex matrices such as wastewater and leachate.
| + | <br clear="right"/> |
| − | Site-specific considerations that affect the selection of optimum treatment technologies for a given site include water chemistry, required flow rate, treatment criteria, waste residual generation, residual disposal options, and operational complexity. Treatability studies with site water are highly recommended because every site has different factors that may affect engineering design for these technologies.
| |
| − | | |
| − | ===Membrane Filtration===
| |
| − | [[File: revOsmosisPlant.png | thumb | 500px | Figure 1. A RO municipal drinking water plant in Arizona]]
| |
| − | Given their ability to remove dissolved contaminants at a molecular size level, RO and some NF membranes can be highly effective for PFAS removal. For RO systems (Figure 1), several studies have demonstrated effective removal of perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) (see [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) | PFAS]] for nomenclature) from drinking water with removal rates well above 90%<ref name="Tang2006">Tang, C.Y., Fu, Q.S., Robertson, A.P., Criddle, C.S., and Leckie, J.O., 2006. Use of Reverse Osmosis Membranes to Remove Perfluorooctane Sulfonate (PFOS) from Semiconductor Wastewater. Environmental Science and Technology, 40(23), pp. 7343-7349. [https://doi.org/10.1021/es060831q DOI: 10.1021/es060831q]</ref><ref name="Flores2013">Flores, C., Ventura, F., Martin-Alonso, J., and Caixach, J., 2013. Occurrence of perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) in NE Spanish surface waters and their removal in a drinking water treatment plant that combines conventional and advanced treatments in parallel lines. Science of the Total environment, 461, 618-626. [https://doi.org/10.1016/j.scitotenv.2013.05.026 DOI: 10.1016/j.scitotenv.2013.05.026]</ref><ref name="Appleman2014">Appleman, T.D., Higgins, C.P., Quiñones, O., Vanderford, B.J., Kolstad, C., Zeigler-Holady, J.C., and Dickenson, E.R., 2014. Treatment of poly- and perfluoroalkyl substances in US full-scale water treatment systems. Water Research, 51, pp. 246-255. [https://doi.org/10.1016/j.watres.2013.10.067 DOI: 10.1016/j.watres.2013.10.067]</ref>. RO potable water reuse treatment systems implemented in California have also demonstrated effective PFOS and PFOA removal as reported by the Water Research Foundation (WRF)<ref name="Dickenson2016"/>. Analysis of permeate at both sites referenced by the WRF confirmed that short and long chain PFAS concentrations in the treated water were reduced to levels below test method reporting limits.
| |
| − |
| |
| − | Full-scale studies using larger effective pore size NF membranes for PFAS removal are limited in number but are promising since NF systems are somewhat less costly than RO and may be nearly as effective in removing PFAS. Recent laboratory or pilot studies have shown good performance of NF membranes<ref name="Steinle-Darling2008">Steinle-Darling, E., and Reinhard, M., 2008. Nanofiltration for Trace Organic Contaminant Removal: Structure, Solution, and Membrane Fouling Effects on the Rejection of Perfluorochemicals. Environmental Science and Technology, 42(14), pp. 5292-5297. [https://doi.org/10.1021/es703207s DOI: 10.1021/es703207s] Free download from: [https://d1wqtxts1xzle7.cloudfront.net/48926882/es703207s20160918-21142-1xmqco5.pdf?1474189169=&response-content-disposition=inline%3B+filename%3DNanofiltration_for_Trace_Organic_Contami.pdf&Expires=1613000850&Signature=N-ZvvjOJX3TSOQzg7od3Q0LulNSZOqqjfummVEUfmiYlC3VasS4FuBHOgY52Xy~7FrKbOLhx0xx8QHdUsR~fbRTMQNXhiqbEslnU2gda2EcZHMMJj0mf-01wIA3jFIywA7IIabmTd3uMUGsIfT1D0PrGY00RmprYIQBoG3Dg~KjoizdfxYfvEgdZw2C~7D47pPiwMSnavZiGuvO0~dbRF8nawL7Prg91xt5BFTNUQQiIrIlMWc4PhVjzE5Su2CUZqnNlYdAW5Ck7B9lKmmVMPiOgz07vFnyp7m-q4UK3woa~aBFW9Wp~hjqN6vfohn8Hocv5oMpZNamhu8vBbPilKw__&Key-Pair-Id=APKAJLOHF5GGSLRBV4ZA Academia].</ref><ref name="Appleman2013">Appleman, T.D., Dickenson, E.R., Bellona, C., and Higgins, C.P., 2013. Nanofiltration and granular activated carbon treatment of perfluoroalkyl acids. Journal of Hazardous Materials, 260, 740-746. [https://doi.org/10.1016/j.jhazmat.2013.06.033 DOI: 10.1016/j.jhazmat.2013.06.033]</ref><ref name="Soriano2017">Soriano, Á., Gorri, D., and Urtiaga, A., 2017. Efficient treatment of perfluorohexanoic acid by nanofiltration followed by electrochemical degradation of the NF concentrate. Water Research, 112, 147-156. [https://doi.org/10.1016/j.watres.2017.01.043 DOI: 10.1016/j.watres.2017.01.043] [[Media: Soriano2017.pdf | Author’s Manuscript.]]</ref><ref name="Zeng2017">Zeng, C., Tanaka, S., Suzuki, Y., Yukioka, S., and Fujii, S., 2017. Rejection of Trace Level Perfluorohexanoic Acid (PFHxA) in Pure Water by Loose Nanofiltration Membrane. Journal of Water and Environment Technology, 15(3), pp. 120-127. [https://doi.org/10.2965/jwet.16-072 DOI: 10.2965/jwet.16-072] Free download from: [https://www.jstage.jst.go.jp/article/jwet/15/3/15_16-072/_pdf J-STAGE]</ref><ref name="Wang2018">Wang, J., Wang, L., Xu, C., Zhi, R., Miao, R., Liang, T., Yue, X., Lv, Y. and Liu, T., 2018. Perfluorooctane sulfonate and perfluorobutane sulfonate removal from water by nanofiltration membrane: The roles of solute concentration, ionic strength, and macromolecular organic foulants. Chemical Engineering Journal, 332, pp. 787-797. [https://doi.org/10.1016/j.cej.2017.09.061 DOI: 10.1016/j.cej.2017.09.061]</ref>.
| |
| − | | |
| − | Although membrane RO and NF processes are generally capable of providing uniform removal rates relative to short and long chain PFAS compounds (see [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) | PFAS]] for nomenclature), other aspects of these treatment technologies are more challenging:
| |
| − | | |
| − | * Membranes must be flushed and cleaned periodically, such that overall water recovery rates (process water volumes consumed, wasted, and lost vs. treated water volumes produced) are much lower than those for GAC and IX processes. Membrane fouling can be slowed or avoided depending on operating conditions, membrane modifications, and feed modifications<ref name="LeRoux2005">Le Roux, I., Krieg, H.M., Yeates, C.A. and Breytenbach, J.C., 2005. Use of chitosan as an antifouling agent in a membrane bioreactor. Journal of Membrane Science, 248(1-2), pp. 127-136. [https://doi.org/10.1016/j.memsci.2004.10.005 DOI: 10.1016/j.memsci.2004.10.005]</ref>. Typically, 70-90% of the water supplied into a membrane RO process is recoverable as treated water. The remaining 10-30% is reject containing approximately 4 to 8 times the initial PFAS concentration (depending on recovery rate).
| |
| − | | |
| − | * These cleaning and flushing processes create a continuous liquid waste stream, which periodically includes harsh membrane cleaning chemicals as well as a continuous flow of concentrated membrane reject chemicals (i.e., PFAS) that must be properly managed and disposed of. Management often includes further treatment to remove PFAS from the liquid waste.
| |
| − | | |
| − | * RO and NF systems are inherently more expensive and complicated systems to implement, operate, and maintain compared to adsorption processes. Treatment system operator certification and process monitoring requirements are correspondingly markedly higher for RO and NF than they are for GAC and IX.
| |
| − | | |
| − | * Water feed pressures required to drive flow through membrane RO and NF processes are considerably higher than those involved with GAC and IX processes. This results in reduced process efficiency and higher pumping and electrical operating costs.
| |
| − | | |
| − | * Membrane systems can also be subject to issues with irreversible membrane fouling, clogging, and scaling or other physical membrane damage and failures. Additional water pretreatment and higher levels of monitoring and maintenance are then required, further adding to the higher costs of such systems.
| |
| − | | |
| − | ===Activated Carbon Adsorption===
| |
| − | [[File: GAChouse.JPG | thumb| 500px | Figure 2. Typical private water supply well GAC installation for removal PFAS. Pressure gages and sample ports located before the first (or lead) vessel, at the midpoint, and after the second (or lag) vessel allow monitoring for pressure drop due to fouling and for contaminant breakthrough.]]
| |
| − | Activated carbon is a form of carbon processed to have small pores that increase the surface area available for adsorption of constituents from water. Activated carbon is derived from many source materials, including coconut shells, wood, lignite, and bituminous coal. Different types of activated carbon base materials have varied adsorption characteristics such that some may be better suited to removing certain contaminant compounds than others. Results from laboratory testing, pilot evaluations, and full-scale system operations suggest that bituminous coal-based GAC is generally the best performing carbon for PFAS removal<ref name="McNamara2018">McNamara, J.D., Franco, R., Mimna, R., and Zappa, L., 2018. Comparison of Activated Carbons for Removal of Perfluorinated Compounds from Drinking Water. Journal‐American Water Works Association, 110(1), pp. E2-E14. [https://doi.org/10.5942/jawwa.2018.110.0003 DOI: 10.5942/jawwa.2018.110.0003]</ref><ref name="Westreich2018">Westreich, P., Mimna, R., Brewer, J., and Forrester, F., 2018. The removal of short‐chain and long‐chain perfluoroalkyl acids and sulfonates via granular activated carbons: A comparative column study. Remediation Journal, 29(1), pp. 19-26. [https://doi.org/10.1002/rem.21579 DOI: 10.1002/rem.21579]</ref>.
| |
| − | | |
| − | The removal efficiency of individual PFAS compounds using GAC is a function of both the PFAS functional group (carboxylic acid versus sulfonic acid) and also the perfluoro-carbon chain length<ref name="McCleaf2017">McCleaf, P., Englund, S., Östlund, A., Lindegren, K., Wiberg, K., and Ahrens, L., 2017. Removal efficiency of multiple poly-and perfluoroalkyl substances (PFASs) in drinking water using granular activated carbon (GAC) and anion exchange (AE) column tests. Water Research, 120, pp. 77-87. [https://doi.org/10.1016/j.watres.2017.04.057 DOI: 10.1016/j.watres.2017.04.057]</ref><ref name="Eschauzier2012">Eschauzier, C., Beerendonk, E., Scholte-Veenendaal, P., and De Voogt, P., 2012. Impact of Treatment Processes on the Removal of Perfluoroalkyl Acids from the Drinking Water Production Chain. Environmental Science and Technology, 46(3), pp. 1708-1715. [https://doi.org/10.1021/es201662b DOI: 10.1021/es201662b]</ref>(see [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) | PFAS]] for nomenclature):
| |
| − | * perfluoro-sulfonate acids (PFSAs) are more efficiently removed than perfluoro-carboxylic acids (PFCAs) of the same chain length
| |
| − | * long chain compounds of the same functional group are removed better than the shorter chains
| |
| − | Activated carbon may be applied in drinking water systems as GAC or PAC<ref name="Dudley">Dudley, L.A., Arevalo, E.C., and Knappe, D.R., 2015. Removal of Perfluoroalkyl Substances by PAC Adsorption and Anion Exchange. Water Research Foundation Project #4344. Free download of Executive Summary from: [https://www.waterrf.org/system/files/resource/2019-04/4344_ProjectSummary.pdf Water Research Foundation (Public Plus account)]</ref><ref name="Qian2017">Qian, J., Shen, M., Wang, P., Wang, C., Li, K., Liu, J., Lu, B. and Tian, X., 2017. Perfluorooctane sulfonate adsorption on powder activated carbon: Effect of phosphate (P) competition, pH, and temperature. Chemosphere, 182, pp. 215-222. [https://doi.org/10.1016/j.chemosphere.2017.05.033 DOI: 10.1016/j.chemosphere.2017.05.033]</ref>. GAC has larger granules and is reusable, while PAC has much smaller granules and is not typically reused. PAC has most often been used as a temporary treatment because costs associated with disposal and replacement of the used PAC tend to preclude using it for long-term treatment. A typical GAC installation for a private drinking water well is shown in Figure 2. Contrary to PAC, GAC used to treat PFAS can be reactivated by the manufacturer, driving the PFAS from the GAC and into off-gas. The extracted gas is then treated with thermal oxidation (temperatures often 1200°C to 1400°C). The reactivated GAC is then brought back to the site and reused. Thus, GAC can ultimately be a destructive treatment technology.
| |
| − | | |
| − | [[File: IXcycle.png | thumb | 400px | left | Figure 3. Operational cycle of a packed bed reactor with anion exchange resin beads]]
| |
| − | | |
| − | ===Anion Exchange===
| |
| − | Anion exchange has also been demonstrated for the adsorption of PFAS, and published results note higher sorption per pound than GAC<ref name="McCleaf2017"/><ref name=" Senevirathna2010">Senevirathna, S.T.M.L.D., Tanaka, S., Fujii, S., Kunacheva, C., Harada, H., Shivakoti, B.R., and Okamoto, R., 2010. A comparative study of adsorption of perfluorooctane sulfonate (PFOS) onto granular activated carbon, ion-exchange polymers and non-ion-exchange polymers. Chemosphere, 80(6), pp. 647-651. [https://doi.org/10.1016/j.chemosphere.2010.04.053 DOI: 10.1016/j.chemosphere.2010.04.053] Free download from: [https://www.researchgate.net/profile/Chinagarn_Kunacheva/publication/44672056_A_comparative_study_of_adsorption_of_perfluorooctane_sulfonate_PFOS_onto_granular_activated_carbon_ion-exchange_polymers_and_non-ion-exchange_polymers/links/5a3380510f7e9b2a288a2b21/A-comparative-study-of-adsorption-of-perfluorooctane-sulfonate-PFOS-onto-granular-activated-carbon-ion-exchange-polymers-and-non-ion-exchange-polymers.pdf ResearchGate]</ref><ref name="Woodard2017">Woodard, S., Berry, J., and Newman, B., 2017. Ion exchange resin for PFAS removal and pilot test comparison to GAC. Remediation Journal, 27(3), pp. 19-27. [https://doi.org/10.1002/rem.21515 DOI: 10.1002/rem.21515]</ref>. The higher capacity is believed to be due to combined hydrophobic and ion exchange adsorption mechanisms, whereas GAC mainly relies on hydrophobic attraction. Anion exchange resins can be highly selective, or they can also remove other contaminants based on design requirements and water chemistry. Resins have greater affinity for PFAS subgroup PFSA than for PFCA, and affinity increases with carbon chain length.
| |
| − | [[Wikipedia: Ion-exchange resin | Anion exchange resins]] are a viable alternative to GAC for ''ex situ'' treatment of PFAS anions, and several venders sell resins capable of removing PFAS. Resins available for treating PFAS include regenerable resins that can be used multiple times (Figure 3) and single-use resins that must be disposed or destroyed after use<ref name=" Senevirathna2010"/>. Regenerable resins generate a solvent and brine solution, which is distilled to recover the solvent prior to the brine being adsorbed onto a small quantity of GAC or resin for ultimate disposal. This use of one treatment technology (GAC, IX) to support another (RO) is sometimes referred to as a “treatment train” approach. Single-use resins can be more fully exhausted than regenerable resins can and may be a more cost-effective solution for low concentration PFAS contamination, while regenerable resins may be more cost effective for higher concentration contamination.
| |
| − | | |
| − | ==Developing PFAS Treatment Technologies==
| |
| − | {| class="wikitable" style="float:right; margin-left:10px;"
| |
| − | |+ Table 1. Developmental Technologies
| |
| − | |-
| |
| − | ! Stage
| |
| − | ! Separation/Transfer
| |
| − | ! Destructive*
| |
| − | |-
| |
| − | | Developing
| |
| − | |
| |
| − | * Biochar<ref name="Guo2017">Guo, W., Huo, S., Feng, J., and Lu, X., 2017. Adsorption of perfluorooctane sulfonate (PFOS) on corn straw-derived biochar prepared at different pyrolytic temperatures. Journal of the Taiwan Institute of Chemical Engineers, 78, pp. 265-271. [https://doi.org/10.1016/j.jtice.2017.06.013 DOI: 10.1016/j.jtice.2017.06.013]</ref><ref name="Kupryianchyk2016">Kupryianchyk, D., Hale, S.E., Breedveld, G.D., and Cornelissen, G., 2016. Treatment of sites contaminated with perfluorinated compounds using biochar amendment. Chemosphere, 142, pp. 35-40. [https://doi.org/10.1016/j.chemosphere.2015.04.085 DOI: 10.1016/j.chemosphere.2015.04.085] Free download from: [https://www.researchgate.net/profile/Sarah_Hale3/publication/276067521_Treatment_of_sites_contaminated_with_perfluorinated_compounds_using_biochar_amendment/links/5cdbe03b299bf14d959895d9/Treatment-of-sites-contaminated-with-perfluorinated-compounds-using-biochar-amendment.pdf ResearchGate]</ref><ref name="Inyang2017">Inyang, M., and Dickenson, E.R., 2017. The use of carbon adsorbents for the removal of perfluoroalkyl acids from potable reuse systems. Chemosphere, 184, pp. 168-175. [https://doi.org/10.1016/j.chemosphere.2017.05.161 DOI: 10.1016/j.chemosphere.2017.05.161]</ref>
| |
| − | * Modified Zeolites<ref name="Espana2015">Espana, V.A.A., Mallavarapu, M., and Naidu, R., 2015. Treatment technologies for aqueous perfluorooctanesulfonate (PFOS) and perfluorooctanoate (PFOA): A critical review with an emphasis on field testing. Environmental Technology and Innovation, 4, pp. 168-181. [https://doi.org/10.1016/j.eti.2015.06.001 DOI: 10.1016/j.eti.2015.06.001] Free download from: [https://www.researchgate.net/profile/Ravi_Naidu2/publication/341241612_Recent_advances_in_the_analysis_of_per-and_polyfluoroalkyl_substances_PFAS-A_review/links/5eb9e3d892851cd50dab441c/Recent-advances-in-the-analysis-of-per-and-polyfluoroalkyl-substances-PFAS-A-review.pdf ResearchGate]</ref><ref name="CETCO2019">CETCO, 2019. FLUORO-SORB® Adsorbent (product sales brochure). [https://www.mineralstech.com/docs/default-source/performance-materials-documents/cetco/environmental-products/brochures/ps_fluorosorb_am_en_201905_v1.pdf Free download] [[Media: FluoroSorb2019.pdf | Fluoro-Sorb.pdf]]</ref>
| |
| − | * Specialty adsorbents<ref name="Zhang2011">Zhang, Q., Deng, S., Yu, G., and Huang, J., 2011. Removal of perfluorooctane sulfonate from aqueous solution by crosslinked chitosan beads: sorption kinetics and uptake mechanism. Bioresource Technology, 102(3), pp. 2265-2271. [https://doi.org/10.1016/j.biortech.2010.10.040 DOI: 10.1016/j.biortech.2010.10.040]</ref><ref name="Cao2016">Cao, F., Wang, L., Ren, X., and Sun, H., 2016. Synthesis of a perfluorooctanoic acid molecularly imprinted polymer for the selective removal of perfluorooctanoic acid in an aqueous environment. Journal of Applied Polymer Science, 133(15). [https://doi.org/10.1002/app.43192 DOI: 10.1002/app.43192]</ref><ref name="Hu2016">Hu, L., Li, Y., and Zhang, W., 2016. Characterization and application of surface-molecular-imprinted-polymer modified TiO2 nanotubes for removal of perfluorinated chemicals. Water Science and Technology, 74(6), pp. 1417-1425. [https://doi.org/10.2166/wst.2016.321 DOI: 10.2166/wst.2016.321] [[Media: Hu2016.pdf | Free access article.]]</ref>
| |
| − | |
| |
| − | * Electro-oxidation<ref name="Zhang2016">Zhang, C., Tang, J., Peng, C., and Jin, M., 2016. Degradation of perfluorinated compounds in wastewater treatment plant effluents by electrochemical oxidation with Nano-ZnO coated electrodes. Journal of Molecular Liquids, 221, pp. 1145-1150. [https://doi.org/10.1016/j.molliq.2016.06.093 DOI: 10.1016/j.molliq.2016.06.093]</ref><ref name="Urtiaga2015">Urtiaga, A., Fernández-González, C., Gómez-Lavín, S., and Ortiz, I., 2015. Kinetics of the electrochemical mineralization of perfluorooctanoic acid on ultrananocrystalline boron doped conductive diamond electrodes. Chemosphere, 129, pp. 20-26. [https://doi.org/10.1016/j.chemosphere.2014.05.090 DOI: 10.1016/j.chemosphere.2014.05.090] Free download from: [https://d1wqtxts1xzle7.cloudfront.net/39233145/00b7d53b67db54fca5000000.pdf?1445006282=&response-content-disposition=inline%3B+filename%3DKinetics_of_the_electrochemical_minerali.pdf&Expires=1613074964&Signature=Bfvds3n9udSs5F9J00Embf8MRJxumQVJoaj5jEni5mqPnmo2QFGGN3fUvWISkRD1yKfoIhNEDQ0a-ISxfZ9vW9jBTkTjN7ud7aSC3rBelIFdtFasfpEXgPvnqsLfKRTWI5S~QRsHbvK5XbwnKo2VyFAmUcuJUjVFP1PK1kEY9-gB2d-8FwSJWbCAAd83fNWm3zHzbOvdchJ~fjAqlydgq7Pu~AwEeH4Zl1LhcYxajzcenTSiBWmMStfOUpTyETSCpSwF7XKuhKMYGePsit8fAWpxH4dleYWmvOi9Gc9YyTB32qBziOTfeqjhTsA-uqECz9bxyD65voHUW7sEchkrKw__&Key-Pair-Id=APKAJLOHF5GGSLRBV4ZA Academia.edu]</ref><ref name="Schaefer2018">Schaefer, C.E., Choyke, S., Ferguson, P.L., Andaya, C., Burant, A., Maizel, A., Strathmann, T.J. and Higgins, C.P., 2018. Electrochemical Transformations of Perfluoroalkyl Acid (PFAA) Precursors and PFAAs in Groundwater Impacted with Aqueous Film Forming Foams. Environmental Science and Technology, 52(18), pp. 10689-10697. [https://doi.org/10.1021/acs.est.8b02726 DOI: 10.1021/acs.est.8b02726]</ref>
| |
| − | * Heat activated persulfate<ref name="Park2016">Park, S., Lee, L.S., Medina, V. F., Zull, A., and Waisner, S., 2016. Heat-activated persulfate oxidation of PFOA, 6: 2 fluorotelomer sulfonate, and PFOS under conditions suitable for in-situ groundwater remediation. Chemosphere, 145, pp. 376-383. [https://doi.org/10.1016/j.chemosphere.2015.11.097 DOI: 10.1016/j.chemosphere.2015.11.097]</ref>
| |
| − | * Alkaline perozone<ref name="Lin2012">Lin, A.Y.C., Panchangam, S.C., Chang, C.Y., Hong, P.A., and Hsueh, H.F., 2012. Removal of perfluorooctanoic acid and perfluorooctane sulfonate via ozonation under alkaline condition. Journal of Hazardous Materials, 243, pp. 272-277. [https://doi.org/10.1016/j.jhazmat.2012.10.029 DOI: 10.1016/j.jhazmat.2012.10.029]</ref>
| |
| − | * Sonolysis<ref name="Campbell2015">Campbell, T., Hoffmann, M.R., 2015. Sonochemical degradation of perfluorinated surfactants: Power and multiple frequency effects. Separation and Purification Technology, 156(3), pp. 1019-1027. [https://doi.org/10.1016/j.seppur.2015.09.053 DOI: 10.1016/j.seppur.2015.09.053] Free download from: [https://www.researchgate.net/profile/Tammy_Campbell5/publication/282583363_Sonochemical_Degradation_of_Perfluorinated_Surfactants_Power_and_Multiple_Frequency_Effects/links/5bfc40bd92851cbcdd74449b/Sonochemical-Degradation-of-Perfluorinated-Surfactants-Power-and-Multiple-Frequency-Effects.pdf ResearchGate]</ref><ref name="Cheng2010">Cheng, J., Vecitis, C.D., Park, H., Mader, B.T., Hoffmann, M.R., 2010. Sonochemical Degradation of Perfluorooctane Sulfonate (PFOS) and Perfluorooctanoate (PFOA) in Groundwater: Kinetic Effects of Matrix Inorganics. Environmental Science and Technology, 44(1), pp. 445-450. [https://doi.org/10.1021/es902651g DOI: 10.1021/es902651g]</ref><ref name="Gole2018a">Gole, V.L., Sierra-Alvarez, R., Peng, H., Giesy, J.P., Deymier, P., Keswani, M., 2018. Sono-chemical treatment of per- and poly-fluoroalkyl compounds in aqueous film-forming foams by use of a large-scale multi-transducer dual-frequency based acoustic reactor. Ultrasonics Sonochemistry, 45, pp. 213-222. [https://doi.org/10.1016/j.ultsonch.2018.02.014 DOI: 10.1016/j.ultsonch.2018.02.014] [https://www.sciencedirect.com/science/article/pii/S1350417718301937 Open access article.] [[Media: Gole2018a.pdf | Report.pdf]]</ref><ref name="Gole2018b">Gole, V.L., Fishgold, A., Sierra-Alvarez, R., Deymier, P., Keswani, M., 2018. Treatment of perfluorooctane sulfonic acid (PFOS) using a large-scale sonochemical reactor. Separation and Purification Technology, 194, pp. 104-110. [https://doi.org/10.1016/j.seppur.2017.11.009 DOI: 10.1016/j.seppur.2017.11.009]</ref>
| |
| − | * Super Critical Water Oxidation
| |
| − | |-
| |
| − | | Maturing and</br>Demonstrated
| |
| − | |
| |
| − | * Chemical coagulation<ref name="Cornelsen2015">Cornelsen Ltd., 2015. PerfluorAd, PFC Water Treatment Solution (product sales site). [http://www.cornelsen.co.uk/perfluorad-pfc-treatment/ Website]</ref>
| |
| − | * Electrocoagulation<ref name="Wang2016">Wang, Y., Lin, H., Jin, F., Niu, J., Zhao, J., Bi, Y., and Li, Y., 2016. Electrocoagulation mechanism of perfluorooctanoate (PFOA) on a zinc anode: Influence of cathodes and anions. Science of the Total Environment, 557, pp. 542-550. [https://doi.org/10.1016/j.scitotenv.2016.03.114 DOI: 10.1016/j.scitotenv.2016.03.114]</ref>
| |
| − | * Foam fractionation<ref name="Horst2018">Horst, J., McDonough, J., Ross, I., Dickson, M., Miles, J., Hurst, J., and Storch, P., 2018. Water Treatment Technologies for PFAS: The Next Generation. Groundwater Monitoring and Remediation, 38(2), pp. 13-23. [https://doi.org/10.1111/gwmr.12281 DOI: 10.1111/gwmr.12281]</ref><ref name="EPC2017">EPC Media Group Pty Ltd., 2017. OPEC systems delivers PFAS contamination breakthrough. Waste + Water Management Australia, 44(3), 26-27. [https://search.informit.org/doi/10.3316/informit.253699294687114 DOI: 10.3316/informit.253699294687114] ISSN: 1838-7098</ref>
| |
| − | |
| |
| − | * Low temperature plasma<ref name="Stratton2017">Stratton, G.R., Dai, F., Bellona, C.L., Holsen, T.M., Dickenson, E.R., and Mededovic Thagard, S., 2017. Plasma-Based Water Treatment: Efficient Transformation of Perfluoroalkyl Substances in Prepared Solutions and Contaminated Groundwater. Environmental Science and Technology, 51(3), pp. 1643-1648. [https://doi.org/10.1021/acs.est.6b04215 DOI: 10.1021/acs.est.6b04215]</ref><ref name="Singh2019">Singh, R.K., Multari, N., Nau-Hix, C., Anderson, R.H., Richardson, S.D., Holsen, T.M. and Mededovic Thagard, S., 2019. Rapid Removal of Poly- and Perfluorinated Compounds from Investigation-Derived Waste (IDW) in a Pilot-Scale Plasma Reactor. Environmental Science and Technology, 53(19), pp. 11375-11382. [https://doi.org/10.1021/acs.est.9b02964 DOI: 10.1021/acs.est.9b02964]</ref>
| |
| − | |-
| |
| − | | colspan="3" style="background:white;" | * There are several other destructive technologies such as alternative oxidants, and activation</br>methods of oxidants, but for the purpose of this article, the main categories are presented here.
| |
| − | |}
| |
| − | Numerous separation and destructive technologies are in the developmental stages of bench-scale testing or limited field-scale demonstrations. Some of these are listed in Table 1:
| |
| − | | |
| − | ==Conclusions==
| |
| − | The well established processes for removing PFAS from water all produce residuals that require management, and it is likely that newer processes under development will also produce some residuals. Often, it is the residuals that limit the usefulness of the process. For instance, RO and NF may currently provide the most complete treatment of water, but the production of a relatively high volume of PFAS-containing liquid reject (the portion of the liquid that retains the contaminants and is “rejected” from the process) limits their application. Often, a second treatment technology such as an adsorbent is required to support the main technology by concentrating or treating the residuals.
| |
| − | As more testing and operational data on adsorbents are generated, it is becoming evident that no adsorbent technology outperforms the others in all cases. Whether GAC, ion exchange or another technology is the most technically efficient and cost effective long term option for a given site depends on influent water geochemistry and contaminant concentrations, treatment standards, co-contaminants, duration of treatment, and required flow rates. New generation adsorbents are rapidly being introduced into the market at “evaluation scale” which may provide advantages over commercially available adsorbents.
| |
| − | Several newer technologies are being evaluated in the lab and in the field which include electro-oxidation, heat-activated persulfate, sonolysis, electrocoagulation, low temperature plasma, super critical water oxidation, and foam fractionation. These and other potential treatments for PFAS are still largely in the developmental stage. Several technologies show promise for improved management of PFAS sites. However, it is unlikely that a single technology will be adequate for full remediation at many sites. A multi-technology treatment train approach may be necessary for effective treatment of this complicated group of compounds.
| |
| − | | |
| − | <br clear="left" /> | |
| | | | |
| | ==References== | | ==References== |
| − |
| |
| | <references /> | | <references /> |
| | | | |
| | ==See Also== | | ==See Also== |
| | + | *[https://serdp-estcp.mil/projects/details/a7e3f7b5-ed82-4591-adaa-6196ff33dd60 ESTCP Project ER20-5031 – In Situ Verification and Quantification of Naturally Occurring Dechlorination Rates in Clays: Demonstrating Processes that Mitigate Back-Diffusion and Plume Persistence] |