|
|
| Line 1: |
Line 1: |
| − | ==PFAS Ex Situ Water Treatment== | + | ==Estimating PCE/TCE Abiotic First-Order Reductive Dechlorination Rate Constants in Clayey Soils Under Anoxic Conditions== |
| − | Well-developed ''ex situ'' treatment technologies applicable to treatment of [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) | perfluoroalkyl and polyfluoroalkyl substances (PFAS)]] in drinking water and non-potable groundwater include membrane filtration (reverse osmosis and nanofiltration), activated carbon adsorption (granular and powdered), and anion exchange. There are also a variety of separation and destructive technologies in developmental stages. Some of these processes may also be applicable to more complex matrices including wastewater and landfill leachate.
| + | The U.S. Department of Defense (DoD) faces many challenges in restoring aquifers at contaminated sites, often due to back-diffusion of tetrachloroethene (PCE) and trichloroethene (TCE) from low-permeability clay zones. The uptake, storage, and subsequent long-term release of these dissolved contaminants from clays are key processes in understanding the longevity, intensity, and risks associated with many persistent chlorinated ethene groundwater plumes. Although naturally occurring abiotic and biotic dechlorination processes in clays may reduce stored contaminant mass and significantly aid natural attenuation, no standardized field method currently exists to verify or quantify these reactions. It is critical to remediation design efforts to demonstrate and validate a cost-effective in situ approach for assessing these dechlorination processes using first-order rate constants. An approach was developed and applied across eight DoD sites to support Remedial Project Managers (RPMs) and regulators in evaluating natural attenuation potential in clay-rich environments. |
| | <div style="float:right;margin:0 0 2em 2em;">__TOC__</div> | | <div style="float:right;margin:0 0 2em 2em;">__TOC__</div> |
| | | | |
| | '''Related Article(s):''' | | '''Related Article(s):''' |
| | | | |
| − | * [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS)]] | + | *[[Monitored Natural Attenuation (MNA)]] |
| − | * [[PFAS Transport and Fate]] | + | *[[Monitored Natural Attenuation (MNA) of Chlorinated Solvents]] |
| − | * [[PFAS Sources]] | + | *[[Monitored Natural Attenuation - Transitioning from Active Remedies]] |
| − | * [[PFAS Soil Remediation Technologies]] | + | *[[Matrix Diffusion]] |
| | + | *[[REMChlor - MD]] |
| | | | |
| − | '''Contributor(s):''' [[Dr. Scott Grieco]] and [[James Hatton]] | + | '''Contributors:''' Dani Tran, Dr. Charles Schaefer, Dr. Charles Werth |
| | | | |
| − | '''Key Resource(s):''' | + | '''Key Resource:''' |
| | + | *Schaefer, C.E, Tran, D., Nguyen, D., Latta, D.E., Werth, C.J., 2025. Evaluating Mineral and In Situ Indicators of Abiotic Dechlorination in Clayey Soils<ref name="SchaeferEtAl2025"/> |
| | | | |
| − | *[https://www.waterrf.org/resource/treatment-mitigation-strategies-poly-and-perfluorinated-chemicals Water Research Foundation (Drinking Water): Treatment Mitigation Strategies for PFAS]<ref name="Dickenson2016">Dickenson, E. and Higgins, C., 2016. Treatment Mitigation Strategies for Poly- and Perfluoroalkyl Substances, Report Number 4322. Water Research Foundation, Denver, Colorado. 123 pages. ISBN 978-1-60573-234-3</ref>
| + | ==Introduction== |
| | + | Cost-effective methods are needed to verify the occurrence of natural dechlorination processes and quantify their dechlorination rates in clays under ambient in situ conditions in order to reliably predict their long-term influence on plume longevity and mass discharge. However, accurately determining these rates is challenging due to slow reaction kinetics, the transient nature of transformation products, and the interplay of biotic and abiotic mechanisms within the clay matrix or at clay-sand interfaces. Tools capable of quantifying these reactions and assessing their role in mitigating plume persistence would be a significant aid for long-term site management. |
| | | | |
| − | *[https://pfas-1.itrcweb.org/12-treatment-technologies/#12_2 Interstate Technical and Regulatory Council: PFAS Liquids Treatment Technologies]<ref name="ITRC2020">Interstate Technology and Regulatory Council (ITRC), 2020. PFAS Technical and Regulatory Guidance Document and Fact Sheets, PFAS-1. PFAS Team, Washington, DC. [https://pfas-1.itrcweb.org/ Website] [[Media: ITRC_PFAS-1.pdf | Report.pdf]]</ref>
| + | For reductive abiotic dechlorination under anoxic conditions, a 1% hydrochloric acid (HCl) extraction of a sample of native clay coupled with X-ray diffraction (XRD) data can be used as a screening level tool to estimate reductive dechlorination rate constants. These rate constants can be inserted into fate and transport models such as [[REMChlor - MD]]<ref>Falta, R., and Wang, W., 2017. A semi-analytical method for simulating matrix diffusion in numerical transport models. Journal of Contaminant Hydrology, 197, pp. 39-49. [https://doi.org/10.1016/j.jconhyd.2016.12.007 doi: 10.1016/j.jconhyd.2016.12.007] [[Media: FaltaWang2017.pdf | Open Access Manuscript]]</ref><ref>Kulkarni, P.R., Adamson, D.T., Popovic, J., Newell, C.J., 2022. Modeling a well-charactized perfluorooctane sulfate (PFOS) source and plume using the REMChlor-MD model to account for matrix diffusion. Journal of Contaminant Hydrology, 247, Article 103986. [https://doi.org/10.1016/j.jconhyd.2022.103986 doi: 10.1016/j.jconhyd.2022.103986] [[Media: KulkarniEtAl2022.pdf | Open Access Manuscript]]</ref> to quantify abiotic dechlorination impacts within clay aquitards on chlorinated solvent plumes. Thus, determination of the abiotic reductive dechlorination rate constant for a particular clayey soil can be readily utilized to provide a more accurate assessment of aquifer cleanup timeframes for groundwater plumes that are being sustained by contaminant back-diffusion. |
| | | | |
| − | *[https://www.sciencedirect.com/science/article/pii/S0301479717307934 Novel treatment technologies for PFAS compounds: A critical review.]<ref name="Kucharzyk2017"> Kucharzyk, K.H., Darlington, R., Benotti, M., Deeb, R. and Hawley, E., 2017. Novel treatment technologies for PFAS compounds: A critical review. Journal of Environmental Management, 204(2), pp. 757-764. [https://doi.org/10.1016/j.jenvman.2017.08.016 DOI: 10.1016/j.jenvman.2017.08.016] Manuscript available from: [https://www.researchgate.net/profile/Katarzyna_kate_Kucharzyk/publication/319125507_Novel_treatment_technologies_for_PFAS_compounds_A_critical_review/links/5a06590b4585157013a3be77/Novel-treatment-technologies-for-PFAS-compounds-A-critical-review.pdf ResearchGate].</ref>
| + | ==Recommended Approach== |
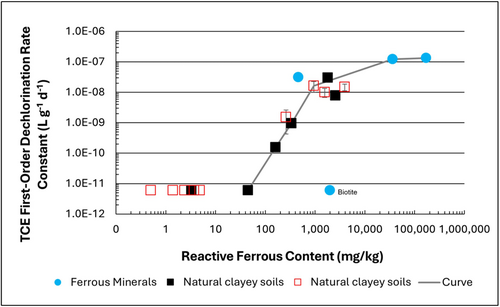
| | + | [[File: TranFig1.png | thumb | 500 px | Figure 1: First-order rate constants for abiotic reductive dechlorination of TCE under anaerobic conditions. Circles are data from Schaefer ''et al.'', 2021<ref>Schaefer, C.E., Ho, P., Berns, E., Werth, C., 2021. Abiotic dechlorination in the presence of ferrous minerals. Journal of Contaminant Hydrology, 241, 103839. [https://doi.org/10.1016/j.jconhyd.2021.103839 doi: 10.1016/j.jconhyd.2021.103839] [[Media: SchaeferEtAl2021.pdf | Open Access Manuscript]]</ref>, filled squares from Schaefer ''et al.'', 2018<ref name="SchaeferEtAl2018"/>, and Schaefer ''et al.'', 2017<ref>Schaefer, C.E., Ho., Gurr, C., Berns, E., Werth, C., 2017. Abiotic dechlorination of chlorinated ethenes in natural clayey soils: impacts of mineralogy and temperature. Journal of Contaminant Hydrology, 206, pp. 10-17. [https://doi.org/10.1016/j.jconhyd.2017.09.007 doi: 10.1016/j.jconhyd.2017.09.007] [[Media: SchaeferEtAl2017.pdf | Open Access Manuscript]]</ref>, and open squares from Schaefer ''et al.'', 2025<ref name="SchaeferEtAl2025"/>. ]] |
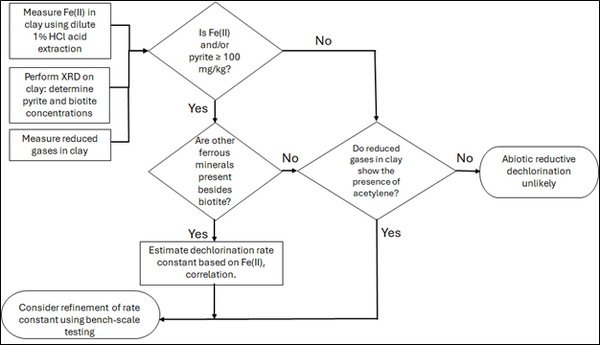
| | + | [[File: TranFig2.png | thumb | 600 px | Figure 2: Flowchart diagram of field screening procedures]] |
| | + | The recommended approach builds upon the methodology and findings of a recent study<ref name="SchaeferEtAl2025">Schaefer, C.E., Tran, D., Nguyen, D., Latta, D.E., Werth, C.J., 2025. Evaluating Mineral and In Situ Indicators of Abiotic Dechlorination in Clayey Soils. Groundwater Monitoring and Remediation, 45(2), pp. 31-39. [https://doi.org/10.1111/gwmr.12709 doi: 10.1111/gwmr.12709]</ref>, emphasizing field-based and analytical techniques to quantify abiotic first-order reductive dechlorination rate constants for PCE and TCE in clayey soils under anoxic conditions. Key components of this evaluation are listed below: |
| | + | #<u>Zone Identification:</u> The focus of the investigation should be to delineate clayey zones adjacent to hydraulically conductive zones. |
| | + | #<u>Ferrous Mineral Quantification:</u> Assess ferrous mineral context in clay via 1% HCl extraction at ambient temperature over a 10-minute interval. |
| | + | #<u>Mineralogical Characterization:</u> Conduct XRD analysis with the specific intent of identifying the presence of pyrite and biotite. |
| | + | #<u>Reduced Gas Analysis:</u> Measurement of reduced gases such as acetylene, ethene, and ethane concentrations in clay samples. Gas-tight sampling devices (e.g., En Core® soil samplers by En Novative Technologies, Inc.) should be used to ensure sample integrity during collection and transport. |
| | | | |
| − | *[https://www.liebertpub.com/doi/abs/10.1089/ees.2016.0233 Degradation and removal methods for perfluoroalkyl and polyfluoroalkyl substances in water]<ref name="Merino2016">Merino, N., Qu, Y., Deeb, R.A., Hawley, E.L., Hoffmann, M.R., and Mahendra, S., 2016. Degradation and Removal Methods for Perfluoroalkyl and Polyfluoroalkyl Substances in Water. Environmental Engineering Science, 33(9), pp. 615-649. [https://doi.org/10.1089/ees.2016.0233 DOI: 10.1089/ees.2016.0233]</ref>
| + | Clay samples should be collected within a few centimeters of the high-permeability interface, with optional additional sampling further inward. For mineralogical analysis, a defined interval may be collected and subsequently subsampled. To preserve sample integrity, exposure to air should be minimized during collection, transport, and handling. Homogenization should occur within an anaerobic chamber, and if subsamples are required for external analysis, they must be shipped in gas-tight, anaerobic containers. |
| | | | |
| − | ==Established PFAS Treatment Technologies==
| + | Estimation of the abiotic reductive first-order rate constant for PCE and TCE is based on the “reactive” ferrous content in the clay. Reactive ferrous content (Fe(II)<sub>r</sub>) is estimated as shown in Equation 1: |
| − | Three technologies are well demonstrated for removal of [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) | PFAS]] from drinking water and non-potable groundwater (as described below):
| |
| − |
| |
| − | * membrane filtration including [[wikipedia: Reverse osmosis | reverse osmosis (RO)]] and [[Wikipedia: Nanofiltration | nanofiltration (NF)]]
| |
| − | * granular [[Wikipedia: Activated carbon | activated carbon]] (GAC) and powdered activated carbon (PAC) adsorption
| |
| − | * [[wikipedia: Ion_exchange | anion exchange (IX)]]
| |
| | | | |
| − | However, these technologies are less demonstrated for removal of PFAS from more complex matrices such as wastewater and leachate.
| + | ::'''Equation 1:''' <big>''Fe(II)<sub><small>r</small></sub> = DA + XRD<sub><small>pyr</small></sub> - XRD<sub><small>biotite</small></sub>''</big> |
| − | Site-specific considerations that affect the selection of optimum treatment technologies for a given site include water chemistry, required flow rate, treatment criteria, waste residual generation, residual disposal options, and operational complexity. Treatability studies with site water are highly recommended because every site has different factors that may affect engineering design for these technologies.
| |
| | | | |
| − | ===Membrane Filtration===
| + | where ''DA'' is the ferrous content from the dilute acid (1% HCl) extraction, ''XRD<sub><small>pyr</small></sub>'' is the pyrite content from XRD analysis, and ''XRD<sub><small>biotite</small></sub>'' is the biotite content from XRD analysis<ref name="SchaeferEtAl2025"/>. |
| − | [[File: revOsmosisPlant.png | thumb | 500px | Figure 1. A RO municipal drinking water plant in Arizona]]
| |
| − | Given their ability to remove dissolved contaminants at a molecular size level, RO and some NF membranes can be highly effective for PFAS removal. For RO systems (Figure 1), several studies have demonstrated effective removal of perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) (see [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) | PFAS]] for nomenclature) from drinking water with removal rates well above 90%<ref name="Tang2006">Tang, C.Y., Fu, Q.S., Robertson, A.P., Criddle, C.S., and Leckie, J.O., 2006. Use of Reverse Osmosis Membranes to Remove Perfluorooctane Sulfonate (PFOS) from Semiconductor Wastewater. Environmental Science and Technology, 40(23), pp. 7343-7349. [https://doi.org/10.1021/es060831q DOI: 10.1021/es060831q]</ref><ref name="Flores2013">Flores, C., Ventura, F., Martin-Alonso, J., and Caixach, J., 2013. Occurrence of perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) in NE Spanish surface waters and their removal in a drinking water treatment plant that combines conventional and advanced treatments in parallel lines. Science of the Total environment, 461, 618-626. [https://doi.org/10.1016/j.scitotenv.2013.05.026 DOI: 10.1016/j.scitotenv.2013.05.026]</ref><ref name="Appleman2014">Appleman, T.D., Higgins, C.P., Quiñones, O., Vanderford, B.J., Kolstad, C., Zeigler-Holady, J.C., and Dickenson, E.R., 2014. Treatment of poly- and perfluoroalkyl substances in US full-scale water treatment systems. Water Research, 51, pp. 246-255. [https://doi.org/10.1016/j.watres.2013.10.067 DOI: 10.1016/j.watres.2013.10.067]</ref>. RO potable water reuse treatment systems implemented in California have also demonstrated effective PFOS and PFOA removal as reported by the Water Research Foundation (WRF)<ref name="Dickenson2016"/>. Analysis of permeate at both sites referenced by the WRF confirmed that short and long chain PFAS concentrations in the treated water were reduced to levels below test method reporting limits.
| |
| − |
| |
| − | Full-scale studies using larger effective pore size NF membranes for PFAS removal are limited in number but are promising since NF systems are somewhat less costly than RO and may be nearly as effective in removing PFAS. Recent laboratory or pilot studies have shown good performance of NF membranes<ref name="Steinle-Darling2008">Steinle-Darling, E., and Reinhard, M., 2008. Nanofiltration for Trace Organic Contaminant Removal: Structure, Solution, and Membrane Fouling Effects on the Rejection of Perfluorochemicals. Environmental Science and Technology, 42(14), pp. 5292-5297. [https://doi.org/10.1021/es703207s DOI: 10.1021/es703207s] Free download from: [https://d1wqtxts1xzle7.cloudfront.net/48926882/es703207s20160918-21142-1xmqco5.pdf?1474189169=&response-content-disposition=inline%3B+filename%3DNanofiltration_for_Trace_Organic_Contami.pdf&Expires=1613000850&Signature=N-ZvvjOJX3TSOQzg7od3Q0LulNSZOqqjfummVEUfmiYlC3VasS4FuBHOgY52Xy~7FrKbOLhx0xx8QHdUsR~fbRTMQNXhiqbEslnU2gda2EcZHMMJj0mf-01wIA3jFIywA7IIabmTd3uMUGsIfT1D0PrGY00RmprYIQBoG3Dg~KjoizdfxYfvEgdZw2C~7D47pPiwMSnavZiGuvO0~dbRF8nawL7Prg91xt5BFTNUQQiIrIlMWc4PhVjzE5Su2CUZqnNlYdAW5Ck7B9lKmmVMPiOgz07vFnyp7m-q4UK3woa~aBFW9Wp~hjqN6vfohn8Hocv5oMpZNamhu8vBbPilKw__&Key-Pair-Id=APKAJLOHF5GGSLRBV4ZA Academia].</ref><ref name="Appleman2013">Appleman, T.D., Dickenson, E.R., Bellona, C., and Higgins, C.P., 2013. Nanofiltration and granular activated carbon treatment of perfluoroalkyl acids. Journal of Hazardous Materials, 260, 740-746. [https://doi.org/10.1016/j.jhazmat.2013.06.033 DOI: 10.1016/j.jhazmat.2013.06.033]</ref><ref name="Soriano2017">Soriano, Á., Gorri, D., and Urtiaga, A., 2017. Efficient treatment of perfluorohexanoic acid by nanofiltration followed by electrochemical degradation of the NF concentrate. Water Research, 112, 147-156. [https://doi.org/10.1016/j.watres.2017.01.043 DOI: 10.1016/j.watres.2017.01.043] [[Media: Soriano2017.pdf | Author’s Manuscript.]]</ref><ref name="Zeng2017">Zeng, C., Tanaka, S., Suzuki, Y., Yukioka, S., and Fujii, S., 2017. Rejection of Trace Level Perfluorohexanoic Acid (PFHxA) in Pure Water by Loose Nanofiltration Membrane. Journal of Water and Environment Technology, 15(3), pp. 120-127. [https://doi.org/10.2965/jwet.16-072 DOI: 10.2965/jwet.16-072] Free download from: [https://www.jstage.jst.go.jp/article/jwet/15/3/15_16-072/_pdf J-STAGE]</ref><ref name="Wang2018">Wang, J., Wang, L., Xu, C., Zhi, R., Miao, R., Liang, T., Yue, X., Lv, Y. and Liu, T., 2018. Perfluorooctane sulfonate and perfluorobutane sulfonate removal from water by nanofiltration membrane: The roles of solute concentration, ionic strength, and macromolecular organic foulants. Chemical Engineering Journal, 332, pp. 787-797. [https://doi.org/10.1016/j.cej.2017.09.061 DOI: 10.1016/j.cej.2017.09.061]</ref>.
| |
| | | | |
| − | Although membrane RO and NF processes are generally capable of providing uniform removal rates relative to short and long chain PFAS compounds (see [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) | PFAS]] for nomenclature), other aspects of these treatment technologies are more challenging:
| + | Abiotic dechlorination is unlikely to contribute to mitigating contaminant back-diffusion when reactive ferrous iron (Fe(II)<sub><small>r</small></sub>) concentrations are below 100 mg/kg (Figure 1). For Fe(II)<sub><small>r</small></sub> above 100 mg/kg, the first-order rate constant for PCE and TCE reductive dechlorination can be estimated using the correlation shown in Figure 1<ref name="SchaeferEtAl2018">Schaefer, C.E., Ho, P., Berns, E., Werth, C., 2018. Mechanisms for abiotic dechlorination of trichloroethene by ferrous minerals under oxic and anoxic conditions in natural sediments. Environmental Science and Technology, 52(23), pp.13747-13755. [https://doi.org/10.1021/acs.est.8b04108 doi: 10.1021/acs.est.8b04108]</ref><ref>Borden, R.C., Cha, K.Y., 2021. Evaluating the impact of back diffusion on groundwater cleanup time. Journal of Contaminant Hydrology, 243, Article 103889. [https://doi.org/10.1016/j.jconhyd.2021.103889 doi: 10.1016/j.jconhyd.2021] [[Media: BordenCha2021.pdf | Open Access Manuscript]]</ref>. The rate constant exhibits a strong positive correlation with the logarithm of reactive Fe(II) content (Pearson’s ''r'' = 0.82), with a slope of 4.7 × 10⁻⁸ L g⁻¹ d⁻¹ (log mg kg⁻¹)⁻¹. |
| | | | |
| − | * Membranes must be flushed and cleaned periodically, such that overall water recovery rates (process water volumes consumed, wasted, and lost vs. treated water volumes produced) are much lower than those for GAC and IX processes. Membrane fouling can be slowed or avoided depending on operating conditions, membrane modifications, and feed modifications<ref name="LeRoux2005">Le Roux, I., Krieg, H.M., Yeates, C.A. and Breytenbach, J.C., 2005. Use of chitosan as an antifouling agent in a membrane bioreactor. Journal of Membrane Science, 248(1-2), pp. 127-136. [https://doi.org/10.1016/j.memsci.2004.10.005 DOI: 10.1016/j.memsci.2004.10.005]</ref>. Typically, 70-90% of the water supplied into a membrane RO process is recoverable as treated water. The remaining 10-30% is reject containing approximately 4 to 8 times the initial PFAS concentration (depending on recovery rate).
| + | Figure 2 presents a decision flowchart designed to evaluate the significance and extent of abiotic reductive dechlorination. By applying Equation 1 to the dilute acid extractable Fe(II) plus measured mineral species data from clay samples, the reactive ferrous iron content (Fe(II)<sub><small>r</small></sub>) can be quantified, enabling a streamlined assessment of the extent to which abiotic processes are contributing to the mitigation of contaminant back-diffusion. |
| | | | |
| − | * These cleaning and flushing processes create a continuous liquid waste stream, which periodically includes harsh membrane cleaning chemicals as well as a continuous flow of concentrated membrane reject chemicals (i.e., PFAS) that must be properly managed and disposed of. Management often includes further treatment to remove PFAS from the liquid waste.
| + | If Fe(II)r is ≥ 100 mg/kg, a first-order dechlorination rate constant can be estimated and subsequently used within a contaminant fate and transport model. However, if acetylene is detected in the clay, even with Fe(II)r less than 100 mg/kg, then bench-scale testing using methods similar to those described in a recent study<ref name="SchaeferEtAl2025"/> is recommended, as such results would likely be inconsistent with those shown in Figure 1, suggesting some other mechanism might be involved, or that the system mineralogy might be more complex than anticipated. Even if Fe(II)r ≥ 100 mg/kg, confirmatory bench-scale testing may be conducted for additional verification and to refine estimation of the abiotic dechlorination rate constant. |
| | | | |
| − | * RO and NF systems are inherently more expensive and complicated systems to implement, operate, and maintain compared to adsorption processes. Treatment system operator certification and process monitoring requirements are correspondingly markedly higher for RO and NF than they are for GAC and IX.
| + | ==Summary and Recommendations== |
| | + | The approach outlined above is intended to serve as a generalized guide for practitioners and site managers to cost-effectively determine the extent to which beneficial abiotic reductive dechlorination reactions are likely occurring in low permeability (e.g., clayey) zones. This approach may be contraindicated if co-contaminants are present. It is currently unclear whether other classes of potentially reactive chemicals, such as trinitrotoluene (TNT) or chlorinated ethanes, could interact competitively with PCE and TCE. |
| | | | |
| − | * Water feed pressures required to drive flow through membrane RO and NF processes are considerably higher than those involved with GAC and IX processes. This results in reduced process efficiency and higher pumping and electrical operating costs.
| + | In addition, it remains unclear how other classes of compounds such as per- and polyfluoroalkyl substances (PFAS) may interact or sorb with ferrous minerals and potentially inhibit abiotic dechlorination reactions. Coupling these recommended activities with conventional site investigation tasks would provide an opportunity to perform many of the up-front screening activities with minimal additional project costs. It is important to note that the guidance proposed herein pertains to particularly low permeability media. Sites with complex or varying lithology, where the mineralogy and/or redox conditions may vary, might require evaluation of multiple samples to provide appropriate site-wide information. |
| | | | |
| − | * Membrane systems can also be subject to issues with irreversible membrane fouling, clogging, and scaling or other physical membrane damage and failures. Additional water pretreatment and higher levels of monitoring and maintenance are then required, further adding to the higher costs of such systems.
| + | <br clear="right"/> |
| − | ===Activated Carbon Adsorption===
| |
| − | [[File: GAChouse.JPG | thumb| 500px | Figure 2. Typical private water supply well GAC installation for removal of VOCs and PFAS. Pressure gages and sample ports installed before the first (or lead) vessel, at the midpoint, and after the second (or lag) vessel allow monitoring for pressure drop due to fouling and for contaminant breakthrough.]]
| |
| − | Activated carbon is a form of carbon processed to have small pores that increase the surface area available for adsorption of constituents from water. Activated carbon is derived from many source materials, including coconut shells, wood, lignite, and bituminous coal. Different types of activated carbon base materials have varied adsorption characteristics such that some may be better suited to removing certain contaminant compounds than others. Results from laboratory testing, pilot evaluations, and full-scale system operations suggest that bituminous coal-based GAC is generally the best performing carbon for PFAS removal<ref name="McNamara2018">McNamara, J.D., Franco, R., Mimna, R., and Zappa, L., 2018. Comparison of Activated Carbons for Removal of Perfluorinated Compounds from Drinking Water. Journal‐American Water Works Association, 110(1), pp. E2-E14. [https://doi.org/10.5942/jawwa.2018.110.0003 DOI: 10.5942/jawwa.2018.110.0003]</ref><ref name="Westreich2018">Westreich, P., Mimna, R., Brewer, J., and Forrester, F., 2018. The removal of short‐chain and long‐chain perfluoroalkyl acids and sulfonates via granular activated carbons: A comparative column study. Remediation Journal, 29(1), pp. 19-26. [https://doi.org/10.1002/rem.21579 DOI: 10.1002/rem.21579]</ref>.
| |
| − | | |
| − | The removal efficiency of individual PFAS compounds using GAC is a function of both the PFAS functional group (carboxylic acid versus sulfonic acid) and also the perfluoro-carbon chain length<ref name="McCleaf2017">McCleaf, P., Englund, S., Östlund, A., Lindegren, K., Wiberg, K., and Ahrens, L., 2017. Removal efficiency of multiple poly-and perfluoroalkyl substances (PFASs) in drinking water using granular activated carbon (GAC) and anion exchange (AE) column tests. Water Research, 120, pp. 77-87. [https://doi.org/10.1016/j.watres.2017.04.057 DOI: 10.1016/j.watres.2017.04.057]</ref><ref name="Eschauzier2012">Eschauzier, C., Beerendonk, E., Scholte-Veenendaal, P., and De Voogt, P., 2012. Impact of Treatment Processes on the Removal of Perfluoroalkyl Acids from the Drinking Water Production Chain. Environmental Science and Technology, 46(3), pp. 1708-1715. [https://doi.org/10.1021/es201662b DOI: 10.1021/es201662b]</ref>
| |
| − | | |
| − | {| class="wikitable" style="float:right; margin-left:10px;"
| |
| − | |+ Table 1. Developmental Technologies
| |
| − | |-
| |
| − | ! Stage
| |
| − | ! Separation/Transfer
| |
| − | ! Destructive*
| |
| − | |-
| |
| − | | Developing
| |
| − | |
| |
| − | * Biochar (20, 21, 22)
| |
| − | * Modified Zeolites (23, 24)
| |
| − | * Specialty adsorbents
| |
| − | |
| |
| − | * Electro-oxidation (32, 33, 34)
| |
| − | * Heat activated persulfate (35)
| |
| − | * Alkaline perozone (36)
| |
| − | * Sonolysis (37, 38, 39, 40)
| |
| − | * Super Critical Water Oxidation
| |
| − | |-
| |
| − | | Maturing and</br>Demonstrated
| |
| − | |
| |
| − | * Chemical coagulation (28)
| |
| − | * Electrocoagulation (29)
| |
| − | * Foam fractionation (30, 31)
| |
| − | |
| |
| − | * Low temperature plasma (41, 42)
| |
| − | |-
| |
| − | | colspan="3" style="background:white;" | * There are several other destructive technologies such as alternative oxidants, and activation</br>methods of oxidants, but for the purpose of this article, the main categories are presented here.
| |
| − | |}
| |
| − | | |
| − | | |
| − | | |
| − | PFAS are a class of highly fluorinated compounds including perfluorooctane sulfonate (PFOS), perfluorooctanoic acid (PFOA), and many other compounds with a variety of industrial and consumer uses. These compounds are often highly resistant to treatment<ref name="Kissa2001">Kissa, Erik, 2001. Fluorinated Surfactants and Repellents: Second Edition. Surfactant Science Series, Volume 97. Marcel Dekker, Inc., CRC Press, New York. 640 pages. ISBN 978-0824704728</ref> and the more mobile compounds are often problematic in groundwater systems<ref name="Backe2013">Backe, W.J., Day, T.C., and Field, J.A., 2013. Zwitterionic, Cationic, and Anionic Fluorinated Chemicals in Aqueous Film Forming Foam Formulations and Groundwater from U.S. Military Bases by Nonaqueous Large-Volume Injection HPLC-MS/MS. Environmental Science and Technology, 47(10), pp. 5226-5234. [https://doi.org/10.1021/es3034999 DOI: 10.1021/es3034999]</ref>. The US EPA has published lifetime drinking water health advisories for the combined concentration of 70 nanograms per liter (ng/L) for two common and recalcitrant PFAS: PFOS, a perfluoroalkyl sulfonic acid (PFSA), and PFOA, a perfluoroalkyl carboxylic acid (PFCA)<ref name="EPApfos2016">US Environmental Protection Agency (EPA), 2016. Drinking Water Health Advisory for Perfluorooctane Sulfonate (PFOS), EPA 822-R-16-004. Office of Water, Health and Ecological Criteria Division, Washington, DC. [https://www.epa.gov/sites/production/files/2016-05/documents/pfos_health_advisory_final-plain.pdf Free download from US EPA] [[Media: USEPA-2016-pfos_health_advisory_final-plain.pdf | Report.pdf]]</ref><ref name="EPApfoa2016">US Environmental Protection Agency (EPA), 2016. Drinking Water Health Advisory for Perfluorooctanoic Acid (PFOA), EPA 822-R-16-005. Office of Water, Health and Ecological Criteria Division, Washington, DC. [https://www.epa.gov/sites/production/files/2016-05/documents/pfoa_health_advisory_final-plain.pdf Free download from US EPA] [[Media: USEPA-2016-pfoa_health_advisory_final-plain.pdf | Report.pdf]]</ref>.(See [[Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS)]] for nomenclature.)
| |
| − | | |
| − | While many of the earliest sites where these compounds were detected in groundwater were manufacturing sites, some recent detections have been attributed to fire training activities associated with aqueous film-forming foams (AFFF). AFFF is the US Department of Defense (DoD) designation for Class B firefighting foam containing PFAS, which is required for fighting fires involving petroleum liquids. Fire training areas and other source areas where AFFF was released at the surface have the potential to be ongoing sources of groundwater contamination<ref name="Houtz2013"/>. (See also [[PFAS Sources]].)
| |
| − | | |
| − | No national soil cleanup standards have been promulgated by the US EPA, although Regional Screening Levels (RSLs) have been calculated and published for perfluorobutane sulfonate (PFBS)<ref name="EPA2020">US Environmental Protection Agency (EPA), 2020. Regional Screening Levels (RSLs) – User's Guide. Washington, DC. [https://www.epa.gov/risk/regional-screening-levels-rsls-users-guide Website]</ref> and data are available to calculate RSLs for PFOA and PFOS<ref name="ITRCwNs2020">Interstate Technology Regulatory Council (ITRC), 2020. PFAS Water and Soil Values Table. PFAS – Per- and Polyfluoroalkyl Substances: PFAS Fact Sheets. [https://pfas-1.itrcweb.org/wp-content/uploads/2020/12/ITRCPFASWaterandSoilValuesTables_NOV-2020-FINAL.xlsx Free download.] [[Media: ITRCPFASWaterandSoilTables2020.xlsx | 2020 Water and Soil Tables (excel file)]]</ref>. Several states have promulgated standards<ref name="AKDEC2020">Alaska Department of Environmental Conservation (AK DEC), 2020. 18 AAC 75, Oil and Other Hazardous Substances Pollution Control. Anchorage, AK. [https://dec.alaska.gov/media/1055/18-aac-75.pdf Free download.] [[Media: AKDEC2020_18aac75.pdf | Report.pdf]]</ref> or screening levels<ref name="MEDEP2018">Maine Department of Environmental Protection (ME DEP), 2018. Maine Remedial Action Guidelines (RAGs) for Sites Contaminated with Hazardous Substances. Augusta, ME. [https://www.maine.gov/dep/spills/publications/guidance/rags/ME-Remedial-Action-Guidelines-10-19-18cc.pdf Free download.] [[Media: MEDEP2018.pdf | Report.pdf]]</ref><ref name="EGLE2020">Michigan Department of Environment, Great Lakes, and Energy (EGLE), 2020. Cleanup Criteria Requirements for Response Activity (Formerly the Part 201 Generic Cleanup Criteria and Screening Levels). Remediation and Redevelopment Division, Lansing, MI. [https://www.michigan.gov/egle/0,9429,7-135-3311_4109_9846-251790--,00.html Website]</ref><ref name="NEDEE2018">Nebraska Department of Energy and Environment (NE DEE), 2018. Voluntary Cleanup Program Remedial Goals, Table A-1: Groundwater and Soil Remediation Goals. Lincoln, NE. [http://www.deq.state.ne.us/Publica.nsf/xsp/.ibmmodres/domino/OpenAttachment/Publica.nsf/D243C2B56E34EA8486256F2700698997/Body/Attach%202-6%20Table%20A-1%20VCP%20LUT%20Sept%202018.pdf Free download.] [[Media: NDEE2018.pdf | Report.pdf]]</ref><ref name="NCDEQ2020">North Carolina Department of Environmental Quality (NC DEQ), 2020. Preliminary Soil Remediation Goals (PSRG) Table. Raleigh, NC. [https://files.nc.gov/ncdeq/risk-based-remediation/1.Combined-Notes-PSRGs.pdf Free download.] [[Media: NCDEQ2020.pdf | Report.pdf]]</ref><ref name="TCEQ2021">Texas Commission on Environmental Quality (TCEQ), 2021. Texas Risk Reduction Program (TRRP), Tier 1 Protective Concentration Levels (PCL) Tables. [http://www.tceq.texas.gov/assets/public/remediation/trrp/2021PCL%20Tables.xlsx Free Download.] [[Media: TRRP2021PCLTables.xlsx | 2021 PCL Tables (excel file)]]</ref> for soil concentrations protective of groundwater, which are several orders of magnitude lower than direct dermal exposure guidelines. These single-digit part per billion criteria will likely drive remedial actions in PFAS source areas in the future. At present, the lack of federally promulgated standards and uncertainty about future standards causes temporary stockpiling of PFAS-impacted soils on sites with soil generated from construction or investigation activities.
| |
| − | | |
| − | ==Soil Treatment==
| |
| − | [[File: DiGuiseppi1w2Fig1.PNG |thumb|600px| Figure 1. A full scale PFAS-impacted soil stabilization project at a military base in Australia. Image courtesy of RemBind™.]]
| |
| − | Addressing recalcitrant contaminants in soil has traditionally been done through containment/capping or excavation and off-site disposal or treatment. Containment/capping may be an acceptable solution for PFAS in some locations. However, containment/capping is not considered ideal given the history of releases from engineered landfills and restrictions on use of land containing capped soils. Innovative treatment approaches for PFAS include stabilization with amendments and thermal treatment.
| |
| − | | |
| − | ===Excavation and Disposal===
| |
| − | Excavation and off-site disposal or treatment of PFAS-impacted soils is the only well-developed treatment technology option and may be acceptable for small quantities of soil, such as those generated during characterization activities (i.e., investigation derived waste, IDW). Disposal in non-hazardous landfills is allowable in most states. However, some landfill operators are choosing to restrict acceptance of PFAS-containing waste and soils as a protection against future liability. In addition, the US EPA and some states are considering or have designated PFOA and PFOS as hazardous substances, which would reduce the number of facilities where disposal of PFAS-contaminated soil would be allowed<ref name="EPA2019">US Environmental Protection Agency (EPA), 2019. EPA’s Per- and Polyfluoroalkyl Substances (PFAS) Action Plan: EPA 823R18004. Washington, DC. [https://www.epa.gov/pfas/epas-pfas-action-plan Website] [[Media: EPA823R18004.pdf | Report.pdf]] [[Media: EPA100K20002.pdf | 2020 Update]]</ref>. Treatment of excavated soils is commonly performed using incineration or other high temperature thermal methods<ref name="ITRC2020"/>. Recent negative publicity regarding incomplete combustion of PFAS in incinerators<ref name="Hogue2020">Cheryl Hogue, 2020. Incineration may spread, not break down PFAS. Chemical and Engineering News, American Chemical Society. [https://cen.acs.org/environment/persistent-pollutants/Incincerators-spread-break-down-PFAS/98/web/2020/04 Website] [[Media: Hogue2020.pdf | Report.pdf]]</ref> has caused some states to ban PFAS incineration<ref name="NYSS2020">New York State Senate, 2020. An ACT prohibiting the incineration of aqueous film-forming foam containing perfluoroalkyl and polyfluoroalkyl substances in certain cities. [https://www.nysenate.gov/legislation/bills/2019/s7880/amendment/b Website] [[Media: NYsenate2020.pdf | Report.pdf]]</ref>.
| |
| − | | |
| − | ===Stabilization===
| |
| − | [[File:DiGuiseppi1w2Fig2.PNG|thumb|600px| Figure 2. A portable infrared thermal treatment unit for PFAS-impacted soils<ref name="DiGuiseppi2019"/>.]]
| |
| − | Various amendments have been manufactured to sorb PFAS to reduce leaching from soil. Although this is a non-destructive approach, stabilization can reduce mass flux from a source area or allow soils to be placed in landfills with reduced potential for leaching. Amendments sorb PFAS through hydrophobic and electrostatic interactions and are applied to soil through ''in situ'' soil mixing or ''ex situ'' stabilization (Figure 1). Effectiveness of amendments varies depending on site conditions, PFAS types present, and mixing conditions<ref name="ITRCwNs2020"/>. Good results have been observed in bench and field scale tests with a variety of cationic clays (natural or chemically modified) and zeolites<ref name="OchoaHerrera2008">Ochoa-Herrera, V., and Sierra-Alvarez, R., 2008. Removal of perfluorinated surfactants by sorption onto granular activated carbon, zeolites and sludge. Chemosphere, 72(10), pp. 1588-1593. [https://doi.org/10.1016/j.chemosphere.2008.04.029 DOI: 10.1016/j.chemosphere.2008.04.029]</ref><ref name="Rattanaoudom2012">Rattanaoudom, R., Visvanathan, C., and Boontanon, S.K., 2012. Removal of Concentrated PFOS and PFOA in Synthetic Industrial Wastewater by Powder Activated Carbon and Hydrotalcite. Journal of Water Sustainability, 2(4), pp. 245-248. [http://www.jwsponline.com/uploadpic/Magazine/pp%20245-258.pdf Open access article.] [[Media: Rattanaoudom2012.pdf | Report.pdf]]</ref><ref name="Ziltek2017">Ziltek, 2017. RemBind: Frequently Asked Questions. [https://static1.squarespace.com/static/5c5503db4d546e22f6d2feb2/t/5c733787f9619ae6c84674c9/1551054727451/RemBind+FAQs.pdf Free download] [[Media: RemBind2017.pdf | Report.pdf]]</ref>. Bench-scale tests have shown that activated carbon sorbents reduce leachability of PFAS from soils<ref name="Du2014">Du, Z., Deng, S., Bei, Y., Huang, Q., Wang, B., Huang, J. and Yu, G., 2014. Adsorption behavior and mechanism of perfluorinated compounds on various adsorbents – A review. Journal of Hazardous Materials, 274, pp. 443-454. [https://doi.org/10.1016/j.jhazmat.2014.04.038 DOI: 10.1016/j.jhazmat.2014.04.038]</ref><ref name="Yu2009">Yu, Q., Zhang, R., Deng, S., Huang, J. and Yu, G., 2009. Sorption of perfluorooctane sulfonate and perfluorooctanoate on activated carbons and resin: Kinetic and isotherm study. Water Research, 43(4), pp. 1150-1158. [https://doi.org/10.1016/j.watres.2008.12.001 DOI: 10.1016/j.watres.2008.12.001]</ref><ref name="Szabo2017">Szabo, J., Hall, J., Magnuson, M., Panguluri, S., and Meiners, G., 2017. Treatment of Perfluorinated Alkyl Substances in Wash Water Using Granular Activated Carbon and Mixed Media, EPA/600/R-17/175. US Environmental Protection Agency (EPA), Washington, DC. [https://cfpub.epa.gov/si/si_public_record_report.cfm?Lab=NHSRC&direntryid=337098 Website] [[Media: EPA600R17175.PDF | Report.pdf]]</ref>. A commercial product developed in Australia ([https://rembind.com/ RemBind™]) combines the cation exchange binding capability of clays, the hydrophobic sorption and [[Wikipedia: Van der Waals force | van der Waals]] attraction of organic material, and the electrostatic interactions of aluminum hydroxide to create a highly effective soil stabilizer. This material has been mixed into soil at 1 to 5% ratio by weight in ''ex situ'' applications and been demonstrated to reduce leachability by greater than 99 percent<ref name="Nolan2015">Nolan, A., Anderson, P., McKay, D., Cartwright, L., and McLean, C., 2015. Treatment of PFCs in Soils, Sediments and Water, WC35. Program and Proceedings, CleanUp Conference 2015. Cooperative Research Centre for Contamination Assessment and Remediation of the Environment (CRC Care), Melbourne, Australia. pp. 374-375. [https://www.crccare.com/files/dmfile/CLEANUP_2015_PROCEEDINGS-web.pdf Free download] [[Media: CRCCare2015.pdf | Report.pdf]]</ref>.
| |
| − | | |
| − | ===Thermal Treatment===
| |
| − | [[File:DiGuiseppi1w2Fig3.PNG|thumb|600px| Figure 3. A full scale PFAS-impacted soil washing plant in Australia<ref name="Grimison2020"/>.]]
| |
| − | ''Incineration:'' Incineration is a well-developed technology for organics destruction, including PFAS-impacted soils. Incineration is generally defined as high temperature (>1,100°C) thermal destruction of waste, and PFAS are thought to mineralize at high temperatures. Generally, incinerators treat off-gasses by thermal oxidation with temperatures as high as 1,400°C, and vaporized combustion products can be captured using condensation and wet scrubbing<ref name="ITRCwNs2020"/>. Some regulatory officials have expressed concern about possible PFAS emissions in off-gas from these incinerators, and the authors are not aware of any published evidence demonstrating complete mineralization of multiple PFAS in incinerators at the time of this posting. In general, incineration is designed to provide “5 nines of destruction” – destruction of 99.999% of the contaminants, although incinerators are not designed to specifically treat PFAS to this standard. In the absence of approved industry standard test methods, the US EPA is developing off-gas/stack testing procedures capable of detecting PFAS at the levels considered to be harmful<ref name="EPA2018">US Environmental Protection Agency (EPA), 2018. PFAS Research and Development, Community Engagement in Fayetteville, North Carolina. [https://www.epa.gov/pfas/pfas-community-engagement-north-carolina-meeting-materials Website] [[Media: EPAFayetteville2018.pdf | Report.pdf]]</ref>.
| |
| − | | |
| − | ''Thermal Desorption:'' Thermal Desorption of PFAS from soil has been demonstrated at the field scale in Australia and the US (Alaska)<ref name="Nolan2015"/> using a rotary kiln operating at temperatures in the range of 900°C or less with treatment times of 10-15 minutes<ref name="Burke2015">Burke, Jill, 2019. Fairbanks incinerator shows promise for cleaning toxic soil. Channel 2-KTUU, October 8. [https://www.ktuu.com/content/news/Fairbanks-incinerator-shows-promise-for-cleaning-toxic-soil-562593631.html Website]</ref>. At these temperatures, some PFAS are mineralized, releasing fluorine that must be captured in off-gas treatment systems. Some PFAS would not be destroyed at these temperatures and therefore must be captured in off-gas treatment systems. Several bench-scale tests have been performed that have narrowed down the optimal temperature for desorption to between 350°C and 400°C<ref name="Hatton2019">Hatton, J., Dasu, K., Richter, R., Fitzpatrick, T., and Higgins, C., 2019. Field Demonstration of Infrared Thermal Treatment of PFAS-impacted Soils from Subsurface Investigations. Strategic Environmental Research and Development Program (SERDP), Project ER18-1603, Alexandria, VA. [https://www.serdp-estcp.org/Program-Areas/Environmental-Restoration/ER18-1603 Website] [[Media: SERDP ER18-1603.pdf | Report.pdf]]</ref><ref name="DiGuiseppi2019">DiGuiseppi, W., Richter, R., and Riggle, M., 2019. Low Temperature Desorption of Per- and Polyfluoroalkyl Substances. The Military Engineer, 111(719), pp. 52-53. Society of American Military Engineers, Washington, DC. [http://online.fliphtml5.com/fedq/sdoo/#p=54 Open access article.] [[Media: DiGuiseppi2019.pdf | Report.pdf]]</ref>. A US Department of Defense (DoD) Strategic Environmental Research and Development Program (SERDP) field-scale demonstration was performed in Oregon, where thermal desorption was conducted at 400°C over several days, and the PFAS were captured on vapor-phase activated carbon and incinerated<ref name="Hatton2019"/>. An ''in situ'' thermal desorption project has been funded under the US DoD’s Environmental Security Technology Certification Program (ESTCP) to demonstrate that vadose zone soil can be heated to the requisite 350°C and held there for the appropriate length of time to desorb and capture PFAS from soil source areas<ref name="Iery2020">Iery, R., 2020. In Situ Thermal Treatment of PFAS in the Vadose Zone. US Department of Defense, Environmental Security Technology Certification Program (ESTCP), Project ER20-5250. [https://www.serdp-estcp.org/Program-Areas/Environmental-Restoration/Contaminated-Groundwater/Emerging-Issues/ER20-5250 Website]</ref>.
| |
| − | | |
| − | ===Soil Washing===
| |
| − | Soil washing has been applied to PFAS in a handful of pilot projects<ref name="Torneman2012">Torneman, N., 2012. Remedial Methods and Strategies for PFCs. Fourth Joint Nordic Meeting on Remediation of Contaminated Sites, NORDROCS 2012, Oslo, Norway. [http://nordrocs.org/wp-content/uploads/2012/09/Session-VI-torsdag-1-Torneman-short-paper.pdf Free download.] [[Media: Torneman2012.pdf | Report.pdf]]</ref><ref name="Toase2018">Toase, D., 2018. Application of enhanced soil washing techniques to PFAS contaminated source zones. Emerging Contaminants Summit 2018, Westminster, Colorado.</ref><ref name="Grimison2018">Grimison, C., Barthelme, S., Nolan, A., Cole, J., Morrell, C., 2018. Integrated Soil and Water System for Treatment of PFAS Impacted Source Areas, 18E138P. Australasian Land and Groundwater Association (ALGA), Sydney, Australia. [https://landandgroundwater.com/media/18E138P_-_Charles_Grimison.pdf Free download.] [[Media: Grimison2018.pdf | Report.pdf]]</ref> and one full-scale implementation in Australia. This approach requires a large-scale engineered plant to handle the various liquid and solid waste streams generated. Soil washing is less suitable for clay-rich soils, where aggregation of the particulates occurs and is difficult to prevent or mitigate. Treatment of the liquid rinse water waste stream is required, which would then rely on conventional water treatment technologies such as granular activated carbon (GAC) or ion exchange. Additionally, in some cases flocculated sludge is generated, which would require treatment or disposal offsite. At present, the only full-scale soil washing demonstration is occurring in Australia, where a vendor has constructed and is operating a 10 million AUD$ treatment plant in anticipation of future treatment of soils generated from remedial actions at Australian Defense installations. Some Australian installations are stockpiling soils due to the lack of cost-effective soil treatment options. According to the vendor, this system generates no solid waste, instead feeding any solids back into the front end of the process for further removal of PFAS<ref name="Grimison2020">Grimison, C., Brookman, I., Hunt, J., and Lucas, J., 2020. Remediation of PFAS-related impacts – ongoing scrutiny and review, Ventia Submission to PFAS Subcommittee of the Joint Standing Committee on Foreign Affairs, Defence and Trade, Australia. [https://www.aph.gov.au/DocumentStore.ashx?id=a209e924-2b7e-4727-bccf-30bef5304bba&subId=691428 Free download.] [[Media: Grimison2020.pdf | Report.pdf]]</ref>.
| |
| − | | |
| − | ==Conclusions==
| |
| − | Several well-developed remedial technologies have been applied to address soil contaminated with PFAS. Unfortunately, none of the available techniques are ideal, with some reducing leachability but leaving the PFAS-impacted soil in place, while others result in destruction of the contaminants but require high energy inputs with associated high cost.
| |
| − | <br clear="left" /> | |
| | | | |
| | ==References== | | ==References== |
| − |
| |
| | <references /> | | <references /> |
| | | | |
| | ==See Also== | | ==See Also== |
| | + | *[https://serdp-estcp.mil/projects/details/a7e3f7b5-ed82-4591-adaa-6196ff33dd60 ESTCP Project ER20-5031 – In Situ Verification and Quantification of Naturally Occurring Dechlorination Rates in Clays: Demonstrating Processes that Mitigate Back-Diffusion and Plume Persistence] |