|
|
| (572 intermediate revisions by the same user not shown) |
| Line 1: |
Line 1: |
| − | ==1,2,3-Trichloropropane (TCP)== | + | ==Munitions Constituents – Sample Extraction and Analytical Techniques== |
| − | [[Wikipedia: 1,2,3-Trichloropropane | 1,2,3-Trichloropropane (TCP)]] is a chlorinated volatile organic compound (CVOC) that has been used in chemical production processes, in agriculture, and as a solvent, resulting in point and non-point source contamination of soil and groundwater. TCP is mobile and highly persistent in soil and groundwater. TCP is not currently regulated at the national level in the United States, but [[Wikipedia: Maximum contaminant level | maximum contaminant levels (MCLs)]] have been developed by some states. Current treatment methods for TCP are limited and can be cost prohibitive. However, some treatment approaches, particularly [[Chemical Reduction (In Situ - ISCR) | ''in situ'' chemical reduction (ISCR)]] with [[Wikipedia: In_situ_chemical_reduction#Zero_valent_metals_%28ZVMs%29 | zero valent zinc (ZVZ)]] and [[Bioremediation - Anaerobic | ''in situ'' bioremediation (ISB)]], have recently been shown to have potential as practical remedies for TCP contamination of groundwater. | + | Munitions Constituents, including [[Wikipedia: Insensitive munition | insensitive munitions]] (IM), are a broad category of compounds which, in areas where manufactured or used, can be found in a variety of environmental matrices (waters, soil, and tissues). This presents an analytical challenge when a variety of these munitions are to be quantified. This article discusses sample extraction methods for each typical sample matrix (high level water, low level water, soil and tissue) as well as the accompanying [[Wikipedia: High-performance liquid chromatography | HPLC]]-UV analytical method for 27 compounds of interest (legacy munitions, insensitive munitions, and surrogates). |
| | + | |
| | <div style="float:right;margin:0 0 2em 2em;">__TOC__</div> | | <div style="float:right;margin:0 0 2em 2em;">__TOC__</div> |
| | | | |
| | '''Related Article(s):''' | | '''Related Article(s):''' |
| − | *[[Bioremediation - Anaerobic | Anaerobic Bioremediation]]
| + | |
| − | *[[Chemical Reduction (In Situ - ISCR) | ''In Situ'' Chemical Reduction (ISCR)]]
| + | *[[Munitions Constituents]] |
| − | *[[Chemical Oxidation (In Situ - ISCO) | ''In Situ'' Chemical Oxidation (ISCO)]] | |
| | | | |
| | '''Contributor(s):''' | | '''Contributor(s):''' |
| − | *[[Dr. Alexandra Salter-Blanc | Alexandra J. Salter-Blanc]]
| + | |
| − | *[[Dr. Paul Tratnyek | Paul G. Tratnyek]]
| + | *Dr. Austin Scircle |
| − | *John Merrill
| |
| − | *Alyssa Saito
| |
| − | *Lea Kane
| |
| − | *Eric Suchomel
| |
| − | *[[Dr. Rula Deeb | Rula Deeb]] | |
| | | | |
| | '''Key Resource(s):''' | | '''Key Resource(s):''' |
| − | *Prospects for Remediation of 1,2,3-Trichloropropane by Natural and Engineered Abiotic Degradation Reactions. Strategic Environmental Research and Development Program (SERDP), Project ER-1457.<ref name="Tratnyek2010">Tratnyek, P.G., Sarathy, V., Salter, A.J., Nurmi, J.T., O’Brien Johnson, G., DeVoe, T., and Lee, P., 2010. Prospects for Remediation of 1,2,3-Trichloropropane by Natural and Engineered Abiotic Degradation Reactions. Strategic Environmental Research and Development Program (SERDP), Project ER-1457. [https://serdp-estcp.org/Program-Areas/Environmental-Restoration/Contaminated-Groundwater/Emerging-Issues/ER-1457/ER-1457/(language)/eng-US Website] [[Media: ER-1457-FR.pdf | Report.pdf]]</ref>
| |
| | | | |
| − | *Verification Monitoring for In Situ Chemical Reduction Using Zero-Valent Zinc, A Novel Technology for Remediation of Chlorinated Alkanes. Strategic Environmental Research and Development Program (SERDP), Project ER-201628.<ref name="Kane2020">Kane, L.Z., Suchomel, E.J., and Deeb, R.A., 2020. Verification Monitoring for In Situ Chemical Reduction Using Zero-Valent Zinc, A Novel Technology for Remediation of Chlorinated Alkanes. Strategic Environmental Research and Development Program (SERDP), Project ER-201628. [https://www.serdp-estcp.org/Program-Areas/Environmental-Restoration/Contaminated-Groundwater/Persistent-Contamination/ER-201628 Website] [[Media: ER-201628.pdf | Report.pdf]]</ref> | + | *[https://www.epa.gov/sites/default/files/2015-07/documents/epa-8330b.pdf USEPA Method 8330B]<ref name= "8330B">United States Environmental Protection Agency (USEPA), 2006. EPA Method 8330B (SW-846) Nitroaromatics, Nitramines, and Nitrate Esters by High Performance Liquid Chromatography (HPLC), Revision 2. [https://www.epa.gov/esam/epa-method-8330b-sw-846-nitroaromatics-nitramines-and-nitrate-esters-high-performance-liquid USEPA Website] [[Media: Epa-2006-method-8330b.pdf | EPA Method 8330b]]</ref> |
| | + | |
| | + | *Methods for simultaneous quantification of legacy and insensitive munition (IM) constituents in aqueous, soil/sediment, and tissue matrices<ref name="CrouchEtAl2020">Crouch, R.A., Smith, J.C., Stromer, B.S., Hubley, C.T., Beal, S., Lotufo, G.R., Butler, A.D., Wynter, M.T., Russell, A.L., Coleman, J.G., Wayne, K.M., Clausen, J.L., Bednar, A.J., 2020. Methods for simultaneous determination of legacy and insensitive munition (IM) constituents in aqueous, soil/sediment, and tissue matrices. Talanta, 217, Article 121008. [https://doi.org/10.1016/j.talanta.2020.121008 doi: 10.1016/j.talanta.2020.121008] [[Media: CrouchEtAl2020.pdf | Open Access Manuscript.pdf]]</ref> |
| | | | |
| | ==Introduction== | | ==Introduction== |
| − | [[File:123TCPFig1.png|thumb|left|Figure 1. Ball and stick representation of the molecular structure of TCP (Salter-Blanc and Tratnyek, unpublished)]]
| + | {| class="wikitable mw-collapsible" style="float:left; margin-right:20px; text-align:center;" |
| − | 1,2,3-Trichloropropane (TCP) (Figure 1) is a man-made chemical that was used in the past primarily as a solvent and extractive agent, as a paint and varnish remover, and as a cleaning and degreasing agent.<ref name="ATSDR2021"> Agency for Toxic Substances and Disease Registry (ATSDR), 2021. Toxicological Profile for 1,2,3-Trichloropropane. Free download from: [https://www.atsdr.cdc.gov/toxprofiles/tp57.pdf ATSDR] [[Media: TCP2021ATSDR.pdf | Report.pdf]]</ref>. Currently, TCP is primarily used in chemical synthesis of compounds such as [[Wikipedia: Polysulfone | polysulfone]] liquid polymers used in the aerospace and automotive industries; [[Wikipedia: Hexafluoropropylene | hexafluoropropylene]] used in the agricultural, electronic, and pharmaceutical industries; [[Wikipedia: Polysulfide | polysulfide]] polymers used as sealants in manufacturing and construction; and [[Wikipedia: 1,3-Dichloropropene | 1,3-dichloropropene]] used in agriculture as a soil fumigant. TCP may also be present in products containing these chemicals as an impurity<ref name="ATSDR2021"/><ref name="CH2M2005">CH2M HILL, 2005. Interim Guidance for Investigating Potential 1,2,3-Trichloropropane Sources in San Gabriel Valley Area 3. [[Media: INTERIM_GUIDANCE_FOR_INVESTIGATING_POTENTIAL_1%2C2%2C3-TRICHLOROPROPANE_SOURCES.pdf | Report.pdf]] [https://cumulis.epa.gov/supercpad/cursites/csitinfo.cfm?id=0902093 Website]</ref>. For example, the 1,2-dichlropropane/1,3-dichloropropene soil fumigant mixture (trade name D-D), which is no longer sold in the United States, contained TCP as an impurity and has been linked to TCP contamination in groundwater<ref name="OkiGiambelluca1987">Oki, D.S. and Giambelluca, T.W., 1987. DBCP, EDB, and TCP Contamination of Ground Water in Hawaii. Groundwater, 25(6), pp. 693-702. [https://doi.org/10.1111/j.1745-6584.1987.tb02210.x DOI: 10.1111/j.1745-6584.1987.tb02210.x]</ref><ref name="CH2M2005"/>. Soil fumigants currently in use which are composed primarily of 1,3-dichloropropene may also contain TCP as an impurity, for instance Telone II has been reported to contain up to 0.17 percent TCP by weight<ref name="Kielhorn2003">Kielhorn, J., Könnecker, G., Pohlenz-Michel, C., Schmidt, S. and Mangelsdorf, I., 2003. Concise International Chemical Assessment Document 56: 1,2,3-Trichloropropane. World Health Organization, Geneva. [http://www.who.int/ipcs/publications/cicad/en/cicad56.pdf Website] [[Media: WHOcicad56TCP.pdf | Report.pdf]]</ref>. | + | |+Table 1. Analyte list with acronyms and CAS numbers. |
| − | | + | |- |
| − | TCP contamination is problematic because it is “reasonably anticipated to be a human carcinogen” based on evidence of carcinogenicity to animals<ref name="NTP2016"> National Toxicology Program, 2016. Report on Carcinogens, 14th ed. U.S. Department of Health and Human Services, Public Health Service. Free download from: [https://ntp.niehs.nih.gov/ntp/roc/content/profiles/trichloropropane.pdf NIH] [[Media: NTP2016trichloropropane.pdf | Report.pdf]]</ref>. Toxicity to humans appears to be high relative to other chlorinated solvents<ref name="Kielhorn2003"/>, suggesting that even low-level exposure to TCP could pose a significant human health risk.
| + | !Compound |
| − | | + | ! Acronym |
| − | ==Environmental Fate==
| + | !CAS Number |
| − | TCP’s fate in the environment is governed by its physical and chemical properties (Table 1). TCP does not adsorb strongly to soil, making it likely to leach into groundwater and exhibit high mobility. In addition, TCP is moderately volatile and can partition from surface water and moist soil into the atmosphere. Because TCP is only slightly soluble and denser than water, it can form a [[Wikipedia: Dense non-aqueous phase liquid | dense non-aqueous phase liquid (DNAPL)]] as observed at the Tyson’s Dump Superfund Site<ref name="USEPA2019"> United States Environmental Protection Agency (USEPA), 2019. Fifth Five-year Review Report, Tyson’s Dump Superfund Site, Upper Merion Township, Montgomery County, Pennsylvania. Free download from: [https://semspub.epa.gov/work/03/2282817.pdf USEPA] [[Media: USEPA2019.pdf | Report.pdf]]</ref>. TCP is generally resistant to aerobic biodegradation, hydrolysis, oxidation, and reduction under naturally occurring conditions making it persistent in the environment<ref name="Tratnyek2010"/>.
| + | |- |
| − | | + | | 1,2-Dinitrobenzene (surrogate) ||'''1,2-DNB (surr.)''' || 528-29-0 |
| − | {| class="wikitable" style="float:right; margin-left:10px;text-align:center;"
| + | |- |
| − | |+Table 1. Physical and chemical properties of TCP<ref name="USEPA2017">United States Environmental Protection Agency (USEPA), 2017. Technical Fact Sheet—1,2,3-Trichloropropane (TCP). EPA Project 505-F-17-007. 6 pp. Free download from: [https://www.epa.gov/sites/production/files/2017-10/documents/ffrrofactsheet_contaminants_tcp_9-15-17_508.pdf USEPA] [[Media: epa_tcp_2017.pdf | Report.pdf]]</ref> | + | | 1,3-Dinitrobenzene || 1,3-DNB || 99-65-0 |
| | + | |- |
| | + | | 1,3,5-Trinitrobenzene || 1,3,5-TNB || 99-35-4 |
| | + | |- |
| | + | | 1,4-Dinitrobenzene || '''1,4-DNB (surr.)''' || 100-25-4 |
| | + | |- |
| | + | | 2-Amino-4,6-dinitrotoluene || 2-Am-4,6-DNT || 35572-78-2 |
| | + | |- |
| | + | | 2-Nitrophenol || '''2-NP''' || 88-75-5 |
| | + | |- |
| | + | | 2-Nitrotoluene || 2-NT || 88-72-2 |
| | + | |- |
| | + | | 2,4-Dinitrophenol || '''2,4-DNP''' || 51-28-5 |
| | + | |- |
| | + | | 2,4-Dinitrotoluene || 2,4-DNT || 121-14-2 |
| | + | |- |
| | + | | 2,4,6-Trinitrophenol || '''Picric Acid (PA)''' || 88-89-1 |
| | + | |- |
| | + | | 2,4,6-Trinitrotoluene || 2,4,6-TNT || 118-96-7 |
| | + | |- |
| | + | | 2,6-Dinitrotoluene || 2,6-DNT || 606-20-2 |
| | + | |- |
| | + | | 3-Nitrotoluene || 3-NT || 99-08-1 |
| | + | |- |
| | + | | 3,5-Dinitroaniline || 3,5-DNA || 618-87-1 |
| | + | |- |
| | + | | 4-Amino-2,6-dinitrotoluene || 4-Am-2,6-DNT || 19406-51-0 |
| | + | |- |
| | + | | 4-Nitrophenol || '''4-NP''' || 100-02-7 |
| | |- | | |- |
| − | !Property
| + | | 4-Nitrotoluene || 4-NT || 99-99-0 |
| − | !Value
| |
| | |- | | |- |
| − | | Chemical Abstracts Service (CAS) Number || 96-18-4 | + | | 2,4-Dinitroanisole || '''DNAN''' || 119-27-7 |
| | |- | | |- |
| − | | Physical Description</br>(at room temperature) || Colorless to straw-colored liquid | + | | Octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine || HMX || 2691-41-0 |
| | |- | | |- |
| − | | Molecular weight (g/mol) || 147.43 | + | | Nitrobenzene || NB || 98-95-3 |
| | |- | | |- |
| − | | Water solubility at 25°C (mg/L)|| 1,750 (slightly soluble) | + | | Nitroglycerine || NG || 55-63-0 |
| | |- | | |- |
| − | | Melting point (°C)|| -14.7 | + | | Nitroguanidine || '''NQ''' || 556-88-7 |
| | |- | | |- |
| − | | Boiling point (°C) || 156.8 | + | | 3-Nitro-1,2,4-triazol-5-one || '''NTO''' || 932-64-9 |
| | |- | | |- |
| − | | Vapor pressure at 25°C (mm Hg) || 3.10 to 3.69 | + | | ''ortho''-Nitrobenzoic acid || '''''o''-NBA (surr.)''' || 552-16-9 |
| | |- | | |- |
| − | | Density at 20°C (g/cm<sup>3</sup>) || 1.3889 | + | | Pentaerythritol tetranitrate || PETN || 78-11-5 |
| | |- | | |- |
| − | | Octanol-water partition coefficient</br>(log''K<sub>ow</sub>'') || 1.98 to 2.27</br>(temperature dependent) | + | | Hexahydro-1,3,5-trinitro-1,3,5-triazine || RDX || 121-82-4 |
| | |- | | |- |
| − | | Organic carbon-water partition coefficient</br>(log''K<sub>oc</sub>'') || 1.70 to 1.99</br>(temperature dependent) | + | | N-Methyl-N-(2,4,6-trinitrophenyl)nitramide || Tetryl || 479-45-8 |
| | |- | | |- |
| − | | Henry’s Law constant at 25°C</br>(atm-m<sup>3</sup>/mol) || 3.17x10<sup>-4</sup><ref name="ATSDR2021"/> to 3.43x10<sup>-4</sup><ref name="LeightonCalo1981">Leighton Jr, D.T. and Calo, J.M., 1981. Distribution Coefficients of Chlorinated Hydrocarbons in Dilute Air-Water Systems for Groundwater Contamination Applications. Journal of Chemical and Engineering Data, 26(4), pp. 382-385. [https://doi.org/10.1021/je00026a010 DOI: 10.1021/je00026a010]</ref> | + | | colspan="3" style="background-color:white;" | Note: Analytes in '''bold''' are not identified by EPA Method 8330B. |
| | |} | | |} |
| | + | [[File: ScircleFig1.png | thumb | 400px | Figure 1. Primary Method labeled chromatograms]] |
| | + | [[File: ScircleFig2.png | thumb | 400px | Figure 2. Secondary Method labeled chromatograms]] |
| | + | The primary intention of the analytical methods presented here is to support the monitoring of legacy and insensitive munitions contamination on test and training ranges, however legacy and insensitive munitions often accompany each other at demilitarization facilities, manufacturing facilities, and other environmental sites. Energetic materials typically appear on ranges as small, solid particulates and due to their varying functional groups and polarities, can partition in various environmental compartments<ref>Walsh, M.R., Temple, T., Bigl, M.F., Tshabalala, S.F., Mai, N. and Ladyman, M., 2017. Investigation of Energetic Particle Distribution from High‐Order Detonations of Munitions. Propellants, Explosives, Pyrotechnics, 42(8), pp. 932-941. [https://doi.org/10.1002/prep.201700089 doi: 10.1002/prep.201700089]</ref>. To ensure that contaminants are monitored and controlled at these sites and to sustainably manage them a variety of sample matrices (surface or groundwater, process waters, soil, and tissues) must be considered. (Process water refers to water used during industrial manufacturing or processing of legacy and insensitive munitions.) Furthermore, additional analytes must be added to existing methodologies as the usage of IM compounds changes and as new degradation compounds are identified. Of note, relatively new IM formulations containing [[Wikipedia: Nitrotriazolone | NTO]], [[Wikipedia: 2,4-Dinitroanisole | DNAN]], and [[Wikipedia: Nitroguanidine | NQ]] are seeing use in [[Wikipedia: IMX-101 | IMX-101]], IMX-104, Pax-21 and Pax-41 (Table 1)<ref>Mainiero, C. 2015. Picatinny Employees Recognized for Insensitive Munitions. U.S. Army, Picatinny Arsenal Public Affairs. [https://www.army.mil/article/148873/picatinny_employees_recognized_for_insensitive_munitions Open Access Press Release]</ref><ref>Frem, D., 2022. A Review on IMX-101 and IMX-104 Melt-Cast Explosives: Insensitive Formulations for the Next-Generation Munition Systems. Propellants, Explosives, Pyrotechnics, 48(1), e202100312. [https://doi.org/10.1002/prep.202100312 doi: 10.1002/prep.202100312]</ref>. |
| | | | |
| − | ==Occurrence==
| + | Sampling procedures for legacy and insensitive munitions are identical and utilize multi-increment sampling procedures found in USEPA Method 8330B Appendix A<ref name= "8330B"/>. Sample hold times, subsampling and quality control requirements are also unchanged. The key differences lie in the extraction methods and instrumental methods. Briefly, legacy munitions analysis of low concentration waters uses a single cartridge reverse phase [[Wikipedia: Solid-phase extraction | SPE]] procedure, and [[Wikipedia: Acetonitrile | acetonitrile]] (ACN) is used for both extraction and [[Wikipedia: Elution | elution]] for aqueous and solid samples<ref name= "8330B"/><ref>United States Environmental Protection Agency (USEPA), 2007. EPA Method 3535A (SW-846) Solid-Phase Extraction (SPE), Revision 1. [https://www.epa.gov/esam/epa-method-3535a-sw-846-solid-phase-extraction-spe USEPA Website] [[Media: epa-3535a.pdf | Method 3535A.pdf]]</ref>. An [[Wikipedia: High-performance_liquid_chromatography#Isocratic_and_gradient_elution | isocratic]] separation via reversed-phase C-18 column with 50:50 methanol:water mobile phase or a C-8 column with 15:85 isopropanol:water mobile phase is used to separate legacy munitions<ref name= "8330B"/>. While these procedures are sufficient for analysis of legacy munitions, alternative solvents, additional SPE cartridges, and a gradient elution are all required for the combined analysis of legacy and insensitive munitions. |
| − | TCP has been detected in approximately 1% of public water supply and domestic well samples tested by the United States Geological Survey. More specifically, TCP was detected in 1.2% of public supply well samples collected between 1993 and 2007 by Toccalino and Hopple<ref name="ToccalinoHopple2010">Toccalino, P.L., Norman, J.E., Hitt, K.J., 2010. Quality of Source Water from Public-Supply Wells in the United States, 1993–2007. Scientific Investigations Report 2010-5024. U.S. Geological Survey. [https://doi.org/10.3133/sir20105024 DOI: 10.3133/sir20105024] Free download from: [https://pubs.er.usgs.gov/publication/sir20105024 USGS] [[Media: Quality_of_source_water_from_public-supply_wells_in_the_United_States%2C_1993-2007.pdf | Report.pdf]]</ref> and 0.66% of domestic supply well samples collected between 1991 and 2004 by DeSimone<ref name="DeSimone2009">DeSimone, L.A., 2009. Quality of Water from Domestic Wells in Principal Aquifers of the United States, 1991–2004. U.S. Geological Survey, Scientific Investigations Report 2008–5227. 139 pp. Free download from: [http://pubs.usgs.gov/sir/2008/5227 USGS] [[Media: DeSimone2009.pdf | Report.pdf]]</ref>. TCP was detected at a higher rate in domestic supply well samples associated with agricultural land-use studies than samples associated with studies comparing primary aquifers (3.5% versus 0.2%)<ref name="DeSimone2009"/>.
| |
| | | | |
| − | ==Regulation==
| + | Previously, analysis of legacy and insensitive munitions required multiple analytical techniques, however the methods presented here combine the two munitions categories resulting in an HPLC-UV method and accompanying extraction methods for a variety of common sample matrices. A secondary HPLC-UV method and a HPLC-MS method were also developed as confirmatory methods. The methods discussed in this article were validated extensively by single-blind round robin testing and subsequent statistical treatment as part of ESTCP [https://serdp-estcp.mil/projects/details/d05c1982-bbfa-42f8-811d-51b540d7ebda ER19-5078]. Wherever possible, the quality control criteria in the Department of Defense Quality Systems Manual for Environmental Laboratories were adhered to<ref>US Department of Defense and US Department of Energy, 2021. Consolidated Quality Systems Manual (QSM) for Environmental Laboratories, Version 5.4. 387 pages. [https://www.denix.osd.mil/edqw/denix-files/sites/43/2021/10/QSM-Version-5.4-FINAL.pdf Free Download] [[Media: QSM-Version-5.4.pdf | QSM Version 5.4.pdf]]</ref>. Analytes included in the methods presented here are found in Table 1. |
| − | The United States Environmental Protection Agency (USEPA) has not established an MCL for TCP, although guidelines and health standards are in place<ref name="USEPA2017"/>. TCP was included in the Contaminant Candidate List 3<ref name="USEPA2009">United States Environmental Protection Agency (US EPA), 2009. Drinking Water Contaminant Candidate List 3-Final. Federal Register 74(194), pp. 51850–51862, Document E9-24287. [https://www.federalregister.gov/documents/2009/10/08/E9-24287/drinking-water-contaminant-candidate-list-3-final Website] [[Media: FR74-194DWCCL3.pdf | Report.pdf]]</ref> and the Unregulated Contaminant Monitoring Rule 3 (UCMR 3)<ref name="USEPA2012">United States Environmental Protection Agency (US EPA), 2012. Revisions to the Unregulated Contaminant Mentoring Regulation (UCMR 3) for Public Water Systems. Federal Register 77(85) pp. 26072-26101. [https://www.federalregister.gov/documents/2012/05/02/2012-9978/revisions-to-the-unregulated-contaminant-monitoring-regulation-ucmr-3-for-public-water-systems Website] [[Media: FR77-85UCMR3.pdf | Report.pdf]]</ref>. The UCMR 3 specified that data be collected on TCP occurrence in public water systems over the period of January 2013 through December 2015 against a reference concentration range of 0.0004 to 0.04 μg/L<ref name="USEPA2017a">United States Environmental Protection Agency (USEPA), 2017. The Third Unregulated Contaminant Monitoring Rule (UCMR 3): Data Summary. EPA 815-S-17-001. [https://www.epa.gov/dwucmr/data-summary-third-unregulated-contaminant-monitoring-rule Website] [[Media: ucmr3-data-summary-january-2017.pdf | Report.pdf]]</ref>. The reference concentration range was determined based on a cancer risk of 10-6 to 10-4 and derived from an oral slope factor of 30 mg/kg-day, which was determined by the EPA’s Integrated Risk Information System<ref name="IRIS2009">USEPA Integrated Risk Information System (IRIS), 2009. 1,2,3-Trichloropropane (CASRN 96-18-4). [https://cfpub.epa.gov/ncea/iris2/chemicalLanding.cfm?substance_nmbr=200 Website] [[Media: TCPsummaryIRIS.pdf | Summary.pdf]]</ref>. Of 36,848 samples collected during UCMR 3, 0.67% exceeded the minimum reporting level of 0.03 µg/L. 1.4% of public water systems had at least one detection over the minimum reporting level, corresponding to 2.5% of the population<ref name="USEPA2017a"/>. While these occurrence percentages are relatively low, the minimum reporting level of 0.03 µg/L is more than 75 times the USEPA-calculated Health Reference Level of 0.0004 µg/L. Because of this, TCP may occur in public water systems at concentrations that exceed the Health Reference Level but are below the minimum reporting level used during UCMR 3 data collection. These analytical limitations and lack of lower-level occurrence data have prevented the USEPA from making a preliminary regulatory determination for TCP<ref name="USEPA2021">USEPA, 2021. Announcement of Final Regulatory Determinations for Contaminants on the Fourth Drinking Water Contaminant Candidate List. Free download from: [https://www.epa.gov/sites/default/files/2021-01/documents/10019.70.ow_ccl_reg_det_4.final_web.pdf USEPA] [[Media: CCL4.pdf | Report.pdf]]</ref>.
| |
| | | | |
| − | Some US states have established their own standards including Hawaii which has established an MCL of 0.6 μg/L<ref name="HDOH2013">Hawaii Department of Health, 2013. Amendment and Compilation of Chapter 11-20 Hawaii Administrative Rules. Free download from: [http://health.hawaii.gov/sdwb/files/2016/06/combodOPPPD.pdf Hawaii Department of Health] [[Media: Amendment_and_Compilation_of_Chapter_11-20_Hawaii_Administrative_Rules.pdf | Report.pdf]]</ref>. California has established an MCL of 0.005 μg/L<ref name="CCR2021">California Code of Regulations, 2021. Section 64444 Maximum Contaminant Levels – Organic Chemicals (22 CA ADC § 64444). [https://govt.westlaw.com/calregs/Document/IA7B3800D18654ABD9E2D24A445A66CB9 Website]</ref>, a notification level of 0.005 μg/L, and a public health goal of 0.0007 μg/L<ref name="OEHHA2009">Office of Environmental Health Hazard Assessment (OEHHA), California Environmental Protection Agency, 2009. Final Public Health Goal for 1,2,3-Trichloropropane in Drinking Water. [https://oehha.ca.gov/water/public-health-goal/final-public-health-goal-123-trichloropropane-drinking-water Website]</ref>, and New Jersey has established an MCL of 0.03 μg/L<ref name="NJAC2020">New Jersey Administrative Code 7:10, 2020. Safe Drinking Water Act Rules. Free download from: [https://www.nj.gov/dep/rules/rules/njac7_10.pdf New Jersey Department of Environmental Protection]</ref>.
| + | The chromatograms produced by the primary and secondary HPLC-UV methods are shown in Figure 1 and Figure 2, respectively. Chromatograms for each detector wavelength used are shown (315, 254, and 210 nm). |
| | | | |
| − | ==Transformation Processes== | + | ==Extraction Methods== |
| − | [[File:123TCPFig2.png|thumb|500px|left|Figure 2. Figure 2. Summary of anticipated primary reaction pathways for degradation of TCP. Oxidation, hydrolysis, and hydrogenolysis are represented by the horizontal arrows. Elimination (dehydrochlorination) and reductive elimination are shown with vertical arrows. [O] represents oxygenation (by oxidation or hydrolysis), [H] represents reduction. Gray indicates products that appear to be of lesser significance<ref name="Tratnyek2010"/>.]] | + | [[File: ScircleFig3.PNG |thumb|400px|Figure 3. Triple cartridge SPE setup]] |
| − | Potential TCP degradation pathways include hydrolysis, oxidation, and reduction (Figure 2). These pathways are expected to be similar overall for abiotic and biotic reactions<ref name="Sarathy2010">Sarathy, V., Salter, A.J., Nurmi, J.T., O’Brien Johnson, G., Johnson, R.L., and Tratnyek, P.G., 2010. Degradation of 1, 2, 3-Trichloropropane (TCP): Hydrolysis, Elimination, and Reduction by Iron and Zinc. Environmental Science and Technology, 44(2), pp.787-793. [https://doi.org/10.1021/es902595j DOI: 10.1021/es902595j]</ref>, but the rates of the reactions (and their resulting significance for remediation) depend on natural and engineered conditions.
| + | [[File: ScircleFig4.PNG |thumb|400px|Figure 4. A flow chart of the soil extraction procedure]] |
| | + | ===High Concentration Waters (> 1 ppm)=== |
| | + | Aqueous samples suspected to contain the compounds of interest at concentrations detectable without any extraction or pre-concentration are suitable for analysis by direct injection. The method deviates from USEPA Method 8330B by adding a pH adjustment and use of MeOH rather than ACN for dilution<ref name= "8330B"/>. The pH adjustment is needed to ensure method accuracy for ionic compounds (like NTO or PA) in basic samples. A solution of 1% HCl/MeOH is added to both acidify and dilute the samples to a final acid concentration of 0.5% (vol/vol) and a final solvent ratio of 1:1 MeOH/H<sub><small>2</small></sub>O. The direct injection samples are then ready for analysis. |
| | | | |
| − | The rate of hydrolysis of TCP is negligible under typical ambient pH and temperature conditions but is favorable at high pH and/or temperature<ref name="Tratnyek2010"/><ref name="Sarathy2010"/>. For example, ammonia gas can be used to raise soil pH and stimulate alkaline hydrolysis of chlorinated propanes including TCP<ref name="Medina2016">Medina, V.F., Waisner, S.A., Griggs, C.S., Coyle, C., and Maxwell, M., 2016. Laboratory-Scale Demonstration Using Dilute Ammonia Gas-Induced Alkaline Hydrolysis of Soil Contaminants (Chlorinated Propanes and Explosives). US Army Engineer Research and Development Center, Environmental Laboratory (ERDC/EL), Report TR-16-10. [http://hdl.handle.net/11681/20312 Website] [[Media: ERDC_EL_TR_16_10.pdf | Report.pdf]]</ref>. [[Thermal Conduction Heating (TCH)]] may also produce favorable conditions for TCP hydrolysis<ref name="Tratnyek2010"/><ref name="Sarathy2010"/>.
| + | ===Low Concentration Waters (< 1 ppm)=== |
| | + | Aqueous samples suspected to contain the compounds of interest at low concentrations require extraction and pre-concentration using solid phase extraction (SPE). The SPE setup described here uses a triple cartridge setup shown in Figure 3. Briefly, the extraction procedure loads analytes of interest onto the cartridges in this order: Strata<sup><small>TM</small></sup> X, Strata<sup><small>TM</small></sup> X-A, and Envi-Carb<sup><small>TM</small></sup>. Then the cartridge order is reversed, and analytes are eluted via a two-step elution, resulting in 2 extracts (which are combined prior to analysis). Five milliliters of MeOH is used for the first elution, while 5 mL of acidified MeOH (2% HCl) is used for the second elution. The particular SPE cartridges used are noncritical so long as cartridge chemistries are comparable to those above. |
| | | | |
| − | ==Treatment Approaches== | + | {| class="wikitable mw-collapsible" style="float:left; margin-right:20px; text-align:center;" |
| − | Compared to more frequently encountered CVOCs such as [[Wikipedia: Trichloroethylene | trichloroethene (TCE)]] and [[Wikipedia: Tetrachloroethylene | tetrachloroethene (PCE)]], TCP is relatively recalcitrant<ref name="Merrill2019">Merrill, J.P., Suchomel, E.J., Varadhan, S., Asher, M., Kane, L.Z., Hawley, E.L., and Deeb, R.A., 2019. Development and Validation of Technologies for Remediation of 1,2,3-Trichloropropane in Groundwater. Current Pollution Reports, 5(4), pp. 228–237. [https://doi.org/10.1007/s40726-019-00122-7 | DOI: 10.1007/s40726-019-00122-7]</ref><ref name="Tratnyek2010"/>. TCP is generally resistant to hydrolysis, bioremediation, oxidation, and reduction under natural conditions<ref name="Tratnyek2010"/>. The moderate volatility of TCP makes air stripping, air sparging, and soil vapor extraction (SVE) less effective compared to other VOCs<ref name="Merrill2019"/>. Despite these challenges, both ''ex situ'' and ''in situ'' treatment technologies exist. ''Ex situ'' treatment processes are relatively well established and understood but can be cost prohibitive. ''In situ'' treatment methods are comparatively limited and less-well developed, though promising field-scale demonstrations of some ''in situ'' treatment technologies have been conducted.
| + | |+Table 2. Primary HPLC-UV mobile phase gradient method concentrations |
| | + | |- |
| | + | | colspan="5" style="background-color:white;"| Method run time = 48 minutes; Column temperature = 25°C<br>Injection volume = 50 μL; Flow rate = 1.0 mL/min<br>Detector wavelengths = 210, 254, and 310 nm |
| | + | |- |
| | + | ! Time<br>(min) |
| | + | ! Reagent Water<br>(%) |
| | + | ! MeOH<br>(%) |
| | + | ! 0.1% TFA/Water<br>(%) |
| | + | ! ACN<br>(%) |
| | + | |- |
| | + | | 0.00 || 89 || 3 || 3 || 5 |
| | + | |- |
| | + | | 2.00 || 89 || 3 || 3 || 5 |
| | + | |- |
| | + | | 2.20 || 52 || 40 || 3 || 5 |
| | + | |- |
| | + | | 12.5 || 52 || 40 || 3 || 5 |
| | + | |- |
| | + | | 19.0 || 57 ||35 || 3 || 5 |
| | + | |- |
| | + | | 28.0 || 48 || 44 || 3 || 5 |
| | + | |- |
| | + | | 32.0 || 48 || 44 || 3 || 5 |
| | + | |- |
| | + | | 44.0 || 32 || 60 || 3 || 5 |
| | + | |- |
| | + | | 44.1 || 89 || 3 || 3 || 5 |
| | + | |- |
| | + | | 48.0 || 89 || 3 || 3 || 5 |
| | + | |} |
| | | | |
| − | ===''Ex Situ'' Treatment=== | + | ===Soils=== |
| − | The most common ''ex situ'' treatment technology for groundwater contaminated with TCP is groundwater extraction and treatment<ref name="SaminJanssen2012">Samin, G. and Janssen, D.B., 2012. Transformation and biodegradation of 1,2,3-trichloropropane (TCP). Environmental Science and Pollution Research International, 19(8), pp. 3067-3078. [https://doi.org/10.1007/s11356-012-0859-3 DOI: 10.1007/s11356-012-0859-3] [[Media: SaminJanssen2012.pdf | Report.pdf]]</ref>. Extraction of TCP is generally effective given its relatively high solubility in water and low degree of partitioning to soil. After extraction, TCP is typically removed by adsorption to granular activated carbon (GAC)<ref name="Merrill2019"/><ref name="CalEPA2017">California Environmental Protection Agency, 2017. Groundwater Information Sheet, 1,2,3-Trichloropropane (TCP). State Water Resources Control Board, Division of Water Quality, Groundwater Ambient Monitoring and Assessment (GAMA) Program, 8 pp. Free download from: [http://www.waterboards.ca.gov/gama/docs/coc_tcp123.pdf California Waterboards] [[Media: CalEPA2017tcp123.pdf | Report.pdf]]</ref>.
| + | Soil collection, storage, drying and grinding procedures are identical to the USEPA Method 8330B procedures<ref name= "8330B"/>; however, the solvent extraction procedure differs in the number of sonication steps, sample mass and solvent used. A flow chart of the soil extraction procedure is shown in Figure 4. Soil masses of approximately 2 g and a sample to solvent ratio of 1:5 (g/mL) are used for soil extraction. The extraction is carried out in a sonication bath chilled below 20 ⁰C and is a two-part extraction, first extracting in MeOH (6 hours) followed by a second sonication in 1:1 MeOH:H<sub><small>2</small></sub>O solution (14 hours). The extracts are centrifuged, and the supernatant is filtered through a 0.45 μm PTFE disk filter. |
| | | | |
| − | TCP contamination in drinking water sources is typically treated using granular activated carbon (GAC)<ref name="Hooker2012">Hooker, E.P., Fulcher, K.G. and Gibb, H.J., 2012. Report to the Hawaii Department of Health, Safe Drinking Water Branch, Regarding the Human Health Risks of 1, 2, 3-Trichloropropane in Tap Water. [https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.269.2485&rep=rep1&type=pdf Free Download] [[Media: Hooker2012.pdf | Report.pdf]]</ref>.
| + | The solvent volume should generally be 10 mL but if different soil masses are required, solvent volume should be 5 mL/g. The extraction results in 2 separate extracts (MeOH and MeOH:H<sub><small>2</small></sub>O) that are combined prior to analysis. |
| | | | |
| − | In California, GAC is considered the best available technology (BAT) for treating TCP, and as of 2017 seven full-scale treatment facilities were using GAC to treat groundwater contaminated with TCP<ref name="CalEPA2017a">California Environmental Protection Agency, 2017. Initial Statement of Reasons 1,2,3-Trichloropropane Maximum Contaminant Level Regulations. Water Resources Control Board, Title 22, California Code of Regulations (SBDDW-17-001). 36 pp. [https://www.waterboards.ca.gov/drinking_water/certlic/drinkingwater/documents/123-tcp/sbddw17_001/isor.pdf Free download]</ref>. Additionally, GAC has been used for over 30 years to treat 60 million gallons per day of TCP-contaminated groundwater in Hawaii<ref name="Babcock2018">Babcock Jr, R.W., Harada, B.K., Lamichhane, K.M., and Tsubota, K.T., 2018. Adsorption of 1, 2, 3-Trichloropropane (TCP) to meet a MCL of 5 ppt. Environmental Pollution, 233, 910-915. [https://doi.org/10.1016/j.envpol.2017.09.085 DOI: 10.1016/j.envpol.2017.09.085]</ref>.
| + | ===Tissues=== |
| | | | |
| − | GAC has a low to moderate adsorption capacity for TCP, which can necessitate larger treatment systems and result in higher treatment costs relative to other organic contaminants<ref name="USEPA2017"/>. Published Freundlich adsorption isotherm parameters<ref name="SnoeyinkSummers1999">Snoeyink, V.L. and Summers, R.S, 1999. Adsorption of Organic Compounds (Chapter 13), In: Water Quality and Treatment, 5th ed., Letterman, R.D., editor. McGraw-Hill, New York, NY. ISBN 0-07-001659-3</ref> indicate that less TCP mass is adsorbed per gram of carbon compared to other volatile organic compounds (VOCs), resulting in increased carbon usage rate and treatment cost. Recent bench-scale studies indicate that subbituminous coal-based GAC and coconut shell-based GAC are the most effective types of GAC for treatment of TCP in groundwater<ref name="Babcock2018"/><ref name="Knappe2017">Knappe, D.R.U., Ingham, R.S., Moreno-Barbosa, J.J., Sun, M., Summers, R.S., and Dougherty, T., 2017. Evaluation of Henry’s Law Constants and Freundlich Adsorption Constants for VOCs. Water Research Foundation Project 4462 Final Report. [https://www.waterrf.org/research/projects/evaluation-henrys-law -constant-and-freundlich-adsorption-constant-vocs Website]</ref>. To develop more economical and effective treatment approaches, further treatability studies with site groundwater (e.g., rapid small-scale column tests) may be needed.
| + | Tissue matrices are extracted by 18-hour sonication using a ratio of 1 gram of wet tissue per 5 mL of MeOH. This extraction is performed in a sonication bath chilled below 20 ⁰C and the supernatant (MeOH) is filtered through a 0.45 μm PTFE disk filter. |
| | | | |
| − | ===''In Situ'' Treatment=== | + | Due to the complexity of tissue matrices, an additional tissue cleanup step, adapted from prior research, can be used to reduce interferences<ref name="RussellEtAl2014">Russell, A.L., Seiter, J.M., Coleman, J.G., Winstead, B., Bednar, A.J., 2014. Analysis of munitions constituents in IMX formulations by HPLC and HPLC-MS. Talanta, 128, pp. 524–530. [https://doi.org/10.1016/j.talanta.2014.02.013 doi: 10.1016/j.talanta.2014.02.013]</ref><ref name="CrouchEtAl2020"/>. The cleanup procedure uses small scale chromatography columns prepared by loading 5 ¾” borosilicate pipettes with 0.2 g activated silica gel (100–200 mesh). The columns are wetted with 1 mL MeOH, which is allowed to fully elute and then discarded prior to loading with 1 mL of extract and collecting in a new amber vial. After the extract is loaded, a 1 mL aliquot of MeOH followed by a 1 mL aliquot of 2% HCL/MeOH is added. This results in a 3 mL silica treated tissue extract. This extract is vortexed and diluted to a final solvent ratio of 1:1 MeOH/H<sub><small>2</small></sub>O before analysis. |
| − | ''In situ'' treatment of TCP to concentrations below current regulatory or advisory levels is difficult to achieve in both natural and engineered systems. However, several ''in situ'' treatment technologies have demonstrated promise for TCP remediation, including chemical reduction by zero-valent metals (ZVMs), chemical oxidation with strong oxidizers, and anaerobic bioremediation<ref name="Merrill2019"/><ref name="Tratnyek2010"/>.
| |
| | | | |
| − | ===''In Situ'' Chemical Reduction (ISCR)=== | + | ==HPLC-UV and HPLC-MS Methods== |
| − | Reduction of TCP under conditions relevant to natural attenuation has been observed to be negligible. Achieving significant degradation rates of TCP requires the addition of a chemical reductant to the contaminated zone<ref name="Merrill2019"/><ref name="Tratnyek2010"/>. Under reducing environmental conditions, some ZVMs have demonstrated the ability to reduce TCP all the way to [[wikipedia:Propene | propene]]. As shown in Figure 2, the desirable pathway for reduction of TCP is the formation of [[Wikipedia: Allyl_chloride | 3-chloro-1-propene (also known as allyl chloride)]] via [[Biodegradation_-_Reductive_Processes#Dihaloelimination | dihaloelimination]], which is then rapidly reduced to propene through [[Wikipedia:Hydrogenolysis | hydrogenolysis]] <ref name="Merrill2019"/><ref name="Tratnyek2010"/><ref name="Torralba-Sanchez2020">Torralba-Sanchez, T.L., Bylaska, E.J., Salter-Blanc, A.J., Meisenheimer, D.E., Lyon, M.A., and Tratnyek, P.G., 2020. Reduction of 1, 2, 3-trichloropropane (TCP): pathways and mechanisms from computational chemistry calculations. Environmental Science: Processes and Impacts, 22(3), 606-616. [https://doi.org/10.1039/C9EM00557A DOI: 10.1039/C9EM00557A]   [[Media: Torralba-Sanchez2020.pdf | Open Access Article]]</ref>. ZVMs including granular zero-valent iron (ZVI), nano ZVI, [[wikipedia: In_situ_chemical_reduction#Bimetallic%20materials | palladized nano ZVI]], and [[wikipedia: In_situ_chemical_reduction#Zero_valent_metals_%28ZVMs%29 | zero-valent zinc (ZVZ)]] have been evaluated by researchers<ref name="Merrill2019"/><ref name="Tratnyek2010"/>.
| + | {| class="wikitable mw-collapsible" style="float:left; margin-right:20px; text-align:center;" |
| − | | + | |+Table 3. Secondary HPLC-UV mobile phase gradient method concentrations |
| − | ZVI is a common reductant used for ISCR and, depending on the form used, has shown variable levels of success for TCP treatment. The Strategic Environmental Research and Development Program (SERDP) Project ER-1457 measured the TCP degradation rates for various forms of ZVI and ZVZ. Nano-scale ZVI and palladized ZVI increased the TCP reduction rate over that of natural attenuation, but the reaction is not anticipated to be fast enough to be useful in typical remediation applications<ref name="Sarathy2010"/>.
| + | |- |
| − | | + | | colspan="5" style="background-color:white;" | Method run time = 43 minutes; Column temperature = 25°C<br>Injection volume = 50 μL; Flow rate = 0.8 mL/min<br>Detector wavelengths = 210, 254, and 310 nm |
| − | Commercial-grade zerovalent zinc (ZVZ) on the other hand is a strong reductant that reduces TCP relatively quickly under a range of laboratory and field conditions to produce propene without significant accumulation of intermediates<ref name="Sarathy2010"/><ref name="Salter-BlancTratnyek2011">Salter-Blanc, A.J. and Tratnyek, P.G., 2011. Effects of Solution Chemistry on the Dechlorination of 1,2,3-Trichloropropane by Zero-Valent Zinc. Environmental Science and Technology, 45(9), pp 4073–4079. [https://doi.org/10.1021/es104081p DOI: 10.1021/es104081p] [[Media: Salter-BlancTratnyek2011.pdf | Open access article]]</ref><ref name="Salter-Blanc2012">Salter-Blanc, A.J., Suchomel, E.J., Fortuna, J.H., Nurmi, J.T., Walker, C., Krug, T., O'Hara, S., Ruiz, N., Morley, T. and Tratnyek, P.G., 2012. Evaluation of Zerovalent Zinc for Treatment of 1,2,3-Trichloropropane‐Contaminated Groundwater: Laboratory and Field Assessment. Groundwater Monitoring and Remediation, 32(4), pp.42-52. [https://doi.org/10.1111/j.1745-6592.2012.01402.x DOI: 10.1111/j.1745-6592.2012.01402.x]</ref><ref name="Merrill2019"/>. Of the ZVMs tested as part of SERDP Project ER-1457, ZVZ had the fastest degradation rates for TCP<ref name="Tratnyek2010"/>. In bench-scale studies, TCP was reduced by ZVZ to propene with 3-chloro-1-propene as the only detectable chlorinated intermediate, which was short-lived and detected only at trace concentrations<ref name="Torralba-Sanchez2020"/>.
| + | |- |
| − | | + | ! Time<br>(min) |
| − | Navy Environmental Sustainability Development to Integration (NESDI) Project 434 conducted bench-scale testing which demonstrated that commercially available ZVZ was effective for treating TCP. Additionally, this project evaluated field-scale ZVZ column treatment of groundwater impacted with TCP at Marine Corps Base Camp Pendleton (MCBCP) in Oceanside, California. This study reported reductions of TCP concentrations by up to 95% which was maintained for at least twelve weeks with influent concentrations ranging from 3.5 to 10 µg/L, without any significant secondary water quality impacts detected<ref name="Salter-Blanc2012"/>.
| + | ! Reagent Water<br>(%) |
| − | | + | ! MeOH<br>(%) |
| − | Following the column study, a 2014 pilot study at MCBCP evaluated direct injection of ZVZ with subsequent monitoring. Direct injection of ZVZ was reportedly effective for TCP treatment, with TCP reductions ranging from 90% to 99% in the injection area. Concentration reduction downgradient of the injection area ranged from 50 to 80%. TCP concentrations have continued to decrease, and reducing conditions have been maintained in the aquifer since injection, demonstrating the long-term efficacy of ZVZ for TCP reduction<ref name="Kane2020"/>.
| + | ! 0.1% TFA/Water<br>(%) |
| − | | + | ! ACN<br>(%) |
| − | Potential ''in situ'' applications of ZVZ include direct injection, as demonstrated by the MCBCP pilot study, and permeable reactive barriers (PRBs). Additionally, ZVZ could potentially be deployed in an ''ex situ'' flow-through reactor, but the economic feasibility of this approach would depend in part on the permeability of the aquifer and in part on the cost of the reactor volumes of ZVZ media necessary for complete treatment.
| + | |- |
| − | | + | | 0.00 || 75 || 10 || 10 || 5 |
| − | ===''In Situ'' Chemical Oxidation (ISCO)===
| + | |- |
| − | Chemical oxidation of TCP with mild oxidants such as permanganate or ozone is ineffective. However, stronger oxidants (e.g. activated peroxide and persulfate) can effectively treat TCP, although the rates are slower than observed for most other organic contaminants<ref name="Tratnyek2010"/><ref name="CalEPA2017"/>. [[Wikipedia: Fenton's reagent | Fenton-like chemistry]] (i.e., Fe(II) activated hydrogen peroxide) has been shown to degrade TCP in the laboratory with half-lives ranging from 5 to 10 hours<ref name="Tratnyek2010"/>, but field-scale demonstrations of this process have not been reported. Treatment of TCP with heat-activated or base-activated persulfate is effective but secondary water quality impacts from high sulfate may be a concern at some locations.
| + | | 2.50 || 75 || 10 || 10 || 5 |
| − | | + | |- |
| − | ===Aerobic Bioremediation===
| + | | 2.60 || 39 || 46 ||10 || 5 |
| − | No naturally occurring microorganisms have been identified that degrade TCP under aerobic conditions<ref name="SaminJanssen2012"/>. Relatively slow aerobic cometabolism by the ammonia oxidizing bacterium [[Wikipedia: Nitrosomonas europaea | Nitrosomonas europaea]] and other populations has been reported<ref name="Vanelli1990">Vannelli, T., Logan, M., Arciero, D.M., and Hooper, A.B., 1990. Degradation of Halogenated Aliphatic Compounds by the Ammonia-Oxidizing Bacterium Nitrosomonas europaea. Applied and Environmental Microbiology, 56(4), pp. 1169–1171. [https://doi.org/10.1128/aem.56.4.1169-1171.1990 DOI: 10.1128/aem.56.4.1169-1171.1990] Free download from: [https://journals.asm.org/doi/epdf/10.1128/aem.56.4.1169-1171.1990 American Society of Microbiology] [[Media: Vannelli1990.pdf | Report.pdf]]</ref><ref name="SaminJanssen2012"/>, and genetic engineering has been used to develop organisms capable of utilizing TCP as a sole carbon source under aerobic conditions<ref name="Bosma2002">Bosma, T., Damborsky, J., Stucki, G., and Janssen, D.B., 2002. Biodegradation of 1,2,3-Trichloropropane through Directed Evolution and Heterologous Expression of a Haloalkane Dehalogenase Gene. Applied and Environmental Microbiology, 68(7), pp. 3582–3587. [https://doi.org/10.1128/AEM.68.7.3582-3587.2002 DOI: 10.1128/AEM.68.7.3582-3587.2002] Free download from: [https://journals.asm.org/doi/epub/10.1128/AEM.68.7.3582-3587.2002 American Society for Microbiology] [[Media: Bosma2002.pdf | Report.pdf]]</ref><ref name="SaminJanssen2012"/><ref name="JanssenStucki2020">Janssen, D. B., and Stucki, G., 2020. Perspectives of genetically engineered microbes for groundwater bioremediation. Environmental Science: Processes and Impacts, 22(3), pp. 487-499. [https://doi.org/10.1039/C9EM00601J DOI: 10.1039/C9EM00601J] Open access article from: [https://pubs.rsc.org/en/content/articlehtml/2020/em/c9em00601j Royal Society of Chemistry] [[Media: JanssenStucki2020.pdf | Report.pdf]]</ref>.
| + | |- |
| − | | + | | 9.00 || 39 || 46 ||10 || 5 |
| − | ===Anaerobic Bioremediation===
| + | |- |
| − | Like other CVOCs, TCP has been shown to undergo biodegradation under anaerobic conditions via reductive dechlorination by [[Wikipedia:Dehalogenimonas | Dehalogenimonas (Dhg)]] species<ref name="Merrill2019"/><ref name="Yan2009">Yan, J., B.A. Rash, F.A. Rainey, and W.M. Moe, 2009. Isolation of novel bacteria within the Chloroflexi capable of reductive dechlorination of 1,2,3-trichloropropane. Environmental Microbiology, 11(4), pp. 833–843. [https://doi.org/10.1111/j.1462-2920.2008.01804.x DOI: 10.1111/j.1462-2920.2008.01804.x]</ref><ref name="Bowman2013">Bowman, K.S., Nobre, M.F., da Costa, M.S., Rainey, F.A., and Moe, W.M., 2013. Dehalogenimonas alkenigignens sp. nov., a chlorinated-alkane-dehalogenating bacterium isolated from groundwater. International Journal of Systematic and Evolutionary Microbiology, 63(Pt_4), pp. 1492-1498. [https://doi.org/10.1099/ijs.0.045054-0 DOI: 10.1099/ijs.0.045054-0] Free access article from: [https://www.microbiologyresearch.org/content/journal/ijsem/10.1099/ijs.0.045054-0?crawler=true Microbiology Society] [[Media: Bowman2013.pdf | Report.pdf]]</ref><ref name="Loffler1997">Loffler, F.E., Champine, J.E., Ritalahti, K.M., Sprague, S.J. and Tiedje, J.M., 1997. Complete Reductive Dechlorination of 1, 2-Dichloropropane by Anaerobic Bacteria. Applied and Environmental Microbiology, 63(7), pp.2870-2875. Free download from: [https://journals.asm.org/doi/pdf/10.1128/aem.63.7.2870-2875.1997 American Society for Micrebiology] [[Medeia: Loffler1997.pdf | Report.pdf]]</ref><ref name="Moe2019">Moe, W.M., Yan, J., Nobre, M.F., da Costa, M.S. and Rainey, F.A., 2009. Dehalogenimonas lykanthroporepellens gen. nov., sp. nov., a reductively dehalogenating bacterium isolated from chlorinated solvent-contaminated groundwater. International Journal of Systematic and Evolutionary Microbiology, 59(11), pp.2692-2697. [https://doi.org/10.1099/ijs.0.011502-0 DOI: 10.1099/ijs.0.011502-0] Free download from: [https://www.microbiologyresearch.org/content/journal/ijsem/10.1099/ijs.0.011502-0?crawler=true Microbiology Society] [[Media: Moe2009.pdf | Report.pdf]]</ref><ref name="SaminJanssen2012"/>. However, the kinetics are slower than for other CVOCs. Bioaugmentation cultures containing Dehalogenimonas (KB-1 Plus, SiREM) are commercially available and have been implemented for remediation of TCP-contaminated groundwater<ref name="Schmitt2017">Schmitt, M., Varadhan, S., Dworatzek, S., Webb, J. and Suchomel, E., 2017. Optimization and validation of enhanced biological reduction of 1,2,3-trichloropropane in groundwater. Remediation Journal, 28(1), pp.17-25. [https://doi.org/10.1002/rem.21539 DOI: 10.1002/rem.21539]</ref>. One laboratory study examined the effect of pH on biotransformation of TCP over a wide range of TCP concentrations (10 to 10,000 µg/L) and demonstrated that successful reduction occurred from a pH of 5 to 9, though optimal conditions were from pH 7 to 9<ref name="Schmitt2017"/>.
| + | | 9.10 || 33.5 || 51.5 || 10 || 5 |
| − | | + | |- |
| − | As with other microbial cultures capable of reductive dechlorination, coordinated amendment with a fermentable organic substrate (e.g. lactate or vegetable oil), also known as biostimulation, creates reducing conditions in the aquifer and provides a source of hydrogen which is required as the primary electron donor for reductive dechlorination.
| + | | 15.00 || 35 || 50 || 10 || 5 |
| − | | + | |- |
| − | A 2016 field demonstration of ''in situ'' bioremediation (ISB) was performed in California’s Central Valley at a former agricultural chemical site with relatively low TCP concentrations (2 µg/L). The site was first biostimulated by injecting amendments of emulsified vegetable oil (EVO) and lactate, which was followed by bioaugmentation with a microbial consortium containing Dhg. After an initial lag period of six months, TCP concentrations decreased to below laboratory detection limits (<0.005 µg/L)<ref name="Schmitt2017"/>.
| + | | 15.10 || 43 || 42 || 10 || 5 |
| − | | + | |- |
| − | The 2016 field demonstration was expanded to full-scale treatment in 2018 with biostimulation and bioaugmentation occurring over several months. The initial TCP concentration in performance monitoring wells ranged from 0.008 to 1.7 µg/L. As with the field demonstration, a lag period of approximately 6 to 8 months was observed before TCP was degraded, after which concentrations declined over fifteen months to non-detectable levels (less than 0.005 µg/L). TCP degradation was associated with increases in Dhg population and propene concentration. Long term monitoring showed that TCP remained at non-detectable levels for at least three years following treatment implementation<ref name="Merrill2019"/>.
| + | | 33.00 || 30 || 55 || 10 || 5 |
| − | | + | |- |
| − | ==Treatment Comparisons and Considerations==
| + | | 33.10 || 75 || 10 || 10 || 5 |
| − | When selecting a technology for TCP treatment, considerations include technical feasibility, ability to treat to regulated levels, potential secondary water quality impacts and relative costs. A comparison of some TCP treatment technologies is provided in Table 2.
| + | |- |
| − | | + | | 43.00 || 75 || 10 || 10 || 5 |
| − | {| class="wikitable" style="float:right; margin-left:10px;text-align:center;" | + | |} |
| − | |+Table 2. Advantages and limitations of TCP treatment technologies<ref name="Kane2020"/> | + | {| class="wikitable mw-collapsible" style="float:right; margin-left:20px; text-align:center;" |
| | + | |+Table 4. Ionization source and detector parameters |
| | + | |- |
| | + | ! Parameter |
| | + | ! Value |
| | + | |- |
| | + | | Ionization Source || APCI |
| | + | |- |
| | + | | Ionization Mode || Negative |
| | + | |- |
| | + | | Drying Gas Temperature (°C) || 350 |
| | + | |- |
| | + | | Vaporizer Temperature (°C) || 325 |
| | + | |- |
| | + | | Drying Gas Flow (L/min) || 4.0 |
| | + | |- |
| | + | | Nebulizer Pressure (psig) || 40 |
| | + | |- |
| | + | | Corona Current (μA) || 10 |
| | + | |- |
| | + | | Capillary Potential (V) || 1500 |
| | + | |- |
| | + | | Mass Range || 40 – 400 |
| | |- | | |- |
| − | ! Technology
| + | | Fragmentor || 100 |
| − | ! Advantages
| |
| − | ! Limitations
| |
| | |- | | |- |
| − | | ZVZ | + | | Gain || 1 |
| − | | style="text-align:left;" | | |
| − | * Can degrade TCP at relatively high and low concentrations
| |
| − | * Faster reaction rates than ZVI
| |
| − | * Material is commercially available
| |
| − | | style="text-align:left;" |
| |
| − | * Higher cost than ZVI
| |
| − | * Difficult to distribute in subsurface ''in situ'' applications
| |
| | |- | | |- |
| − | | Groundwater</br>Extraction and</br>Treatment | + | | Threshold || 0 |
| − | | style="text-align:left;" | | |
| − | * Can cost-effectively capture and treat larger, more dilute</br>groundwater plumes than ''in situ'' technologies
| |
| − | * Well understood and widely applied technology
| |
| − | | style="text-align:left;" |
| |
| − | * Requires construction, operation and maintenance of</br>aboveground treatment infrastructure
| |
| − | * Typical technologies (e.g. GAC) may be expensive due</br>to treatment inefficiencies
| |
| | |- | | |- |
| − | | ZVI | + | | Step Size || 0.20 |
| − | | style="text-align:left;" | | + | |- |
| − | * Can degrade TCP at relatively high and low concentrations
| + | | Speed (μ/sec) || 743 |
| − | * Lower cost than ZVZ
| |
| − | * Material is commercially available
| |
| − | | style="text-align:left;" | | |
| − | * Lower reactivity than ZVZ, therefore may require higher</br>ZVI volumes or thicker PRBs
| |
| − | * Difficult to distribute in subsurface ''in situ'' applications
| |
| | |- | | |- |
| − | | ISCO | + | | Peak Width (min) || 0.06 |
| − | | style="text-align:left;" |
| |
| − | * Can degrade TCP at relatively high and low concentrations
| |
| − | * Strategies to distribute amendments ''in situ'' are well established
| |
| − | * Material is commercially available
| |
| − | | style="text-align:left;" | | |
| − | * Most effective oxidants (e.g., base-activated or heat-activated</br>persulfate) are complex to implement
| |
| − | * Secondary water quality impacts (e.g., high pH, sulfate, </br>hexavalent chromium) may limit ability to implement
| |
| | |- | | |- |
| − | | ''In Situ''</br>Bioremediation | + | | Cycle Time (sec/cycle) || 0.57 |
| − | | style="text-align:left;" | | |
| − | * Can degrade TCP at moderate to high concentrations
| |
| − | * Strategies to distribute amendments ''in situ'' are well established
| |
| − | * Materials are commercially available and inexpensive
| |
| − | | style="text-align:left;" |
| |
| − | * Slower reaction rates than ZVZ or ISCO
| |
| | |} | | |} |
| | + | |
| | + | The Primary HPLC-UV method uses a Phenomenex Synergi 4 µm Hydro-RP column (80Å, 250 x 4.6 mm), or comparable, and is based on both the HPLC method found in USEPA 8330B and previous work<ref name= "8330B"/><ref name="RussellEtAl2014"/><ref name="CrouchEtAl2020"/>. This separation relies on a reverse phase column and uses a gradient elution, shown in Table 2. Depending on the analyst’s needs and equipment availability, the method has been proven to work with either 0.1% TFA or 0.25% FA (vol/vol) mobile phase. Addition of a guard column like a Phenomenex SecurityGuard AQ C18 pre-column guard cartridge can be optionally used. These optional changes to the method have no impact on the method’s performance. |
| | + | |
| | + | The Secondary HPLC-UV method uses a Restek Pinnacle II Biphenyl 5 µm (150 x 4.6 mm) or comparable column and is intended as a confirmatory method. Like the Primary method, this method can use an optional guard column and utilizes a gradient elution, shown in Table 3. |
| | + | |
| | + | For instruments equipped with a mass spectrometer (MS), a secondary MS method is available and was developed alongside the Primary UV method. The method was designed for use with a single quadrupole MS equipped with an atmospheric pressure chemical ionization (APCI) source, such as an Agilent 6120B. A majority of the analytes shown in Table 1 are amenable to this MS method, however nitroglycerine (which is covered extensively in USEPA method 8332) and 2-,3-, and 4-nitrotoluene compounds aren’t compatible with the MS method. MS method parameters are shown in Table 4. |
| | | | |
| | ==Summary== | | ==Summary== |
| − | The relatively high toxicity of TCP has led to the development of health-based drinking water concentration values that are very low. TCP is sometimes present in groundwater and in public water systems at concentrations that exceed these health-based goals. While a handful of states have established MCLs for TCP, US federal regulatory determination is hindered by the lack of low-concentration occurrence data. Because TCP is persistent in groundwater and resistant to typical remediation methods (or costly to treat), specialized strategies may be needed to meet drinking-water-based treatment goals. ''In situ'' chemical reduction (ISCR) with zero valent zinc (ZVZ) and ''in situ'' bioremediation have been demonstrated to be effective for TCP remediation. | + | The extraction methods and instrumental methods in this article build upon prior munitions analytical methods by adding new compounds, combining legacy and insensitive munitions analysis, and expanding usable sample matrices. These methods have been verified through extensive round robin testing and validation, and while the methods are somewhat challenging, they are crucial when simultaneous analysis of both insensitive and legacy munitions is needed. |
| | | | |
| | ==References== | | ==References== |
| Line 178: |
Line 228: |
| | | | |
| | ==See Also== | | ==See Also== |
| − | ATSDR Toxicological Profile: https://www.atsdr.cdc.gov/ToxProfiles/TP.asp?id=912&tid=186
| + | *[[Media: ERDC_TR-21-12.pdf | Preparative, Extraction, and Analytical Methods for Simultaneous Determination of Legacy and Insensitive Munition (IM) Constituents in Aqueous, Soil or Sediment, and Tissue Matrices]] |
| − | | + | *[https://serdp-estcp.mil/focusareas/9f7a342a-1b13-4ce5-bda0-d7693cf2b82d/uxo#subtopics SERDP/ESTCP Focus Areas – UXO – Munitions Constituents] |
| − | EPA Technical Fact Sheet: https://www.epa.gov/sites/production/files/2014-03/documents/ffrrofactsheet_contaminant_tcp_january2014_final.pdf
| + | *[https://denix.osd.mil/edqw/home/ Environmental Data Quality Workgroup] |
| − | | |
| − | Cal/EPA State Water Resources Control Board Groundwater Information Sheet: http://www.waterboards.ca.gov/gama/docs/coc_tcp123.pdf
| |
| − | | |
| − | California Water Boards Fact Sheet: http://www.waterboards.ca.gov/drinking_water/certlic/drinkingwater/documents/123-tcp/123tcp_factsheet.pdf
| |
Munitions Constituents – Sample Extraction and Analytical Techniques
Munitions Constituents, including insensitive munitions (IM), are a broad category of compounds which, in areas where manufactured or used, can be found in a variety of environmental matrices (waters, soil, and tissues). This presents an analytical challenge when a variety of these munitions are to be quantified. This article discusses sample extraction methods for each typical sample matrix (high level water, low level water, soil and tissue) as well as the accompanying HPLC-UV analytical method for 27 compounds of interest (legacy munitions, insensitive munitions, and surrogates).
Related Article(s):
Contributor(s):
Key Resource(s):
- Methods for simultaneous quantification of legacy and insensitive munition (IM) constituents in aqueous, soil/sediment, and tissue matrices[2]
Introduction
Table 1. Analyte list with acronyms and CAS numbers.
| Compound
|
Acronym
|
CAS Number
|
| 1,2-Dinitrobenzene (surrogate) |
1,2-DNB (surr.) |
528-29-0
|
| 1,3-Dinitrobenzene |
1,3-DNB |
99-65-0
|
| 1,3,5-Trinitrobenzene |
1,3,5-TNB |
99-35-4
|
| 1,4-Dinitrobenzene |
1,4-DNB (surr.) |
100-25-4
|
| 2-Amino-4,6-dinitrotoluene |
2-Am-4,6-DNT |
35572-78-2
|
| 2-Nitrophenol |
2-NP |
88-75-5
|
| 2-Nitrotoluene |
2-NT |
88-72-2
|
| 2,4-Dinitrophenol |
2,4-DNP |
51-28-5
|
| 2,4-Dinitrotoluene |
2,4-DNT |
121-14-2
|
| 2,4,6-Trinitrophenol |
Picric Acid (PA) |
88-89-1
|
| 2,4,6-Trinitrotoluene |
2,4,6-TNT |
118-96-7
|
| 2,6-Dinitrotoluene |
2,6-DNT |
606-20-2
|
| 3-Nitrotoluene |
3-NT |
99-08-1
|
| 3,5-Dinitroaniline |
3,5-DNA |
618-87-1
|
| 4-Amino-2,6-dinitrotoluene |
4-Am-2,6-DNT |
19406-51-0
|
| 4-Nitrophenol |
4-NP |
100-02-7
|
| 4-Nitrotoluene |
4-NT |
99-99-0
|
| 2,4-Dinitroanisole |
DNAN |
119-27-7
|
| Octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine |
HMX |
2691-41-0
|
| Nitrobenzene |
NB |
98-95-3
|
| Nitroglycerine |
NG |
55-63-0
|
| Nitroguanidine |
NQ |
556-88-7
|
| 3-Nitro-1,2,4-triazol-5-one |
NTO |
932-64-9
|
| ortho-Nitrobenzoic acid |
o-NBA (surr.) |
552-16-9
|
| Pentaerythritol tetranitrate |
PETN |
78-11-5
|
| Hexahydro-1,3,5-trinitro-1,3,5-triazine |
RDX |
121-82-4
|
| N-Methyl-N-(2,4,6-trinitrophenyl)nitramide |
Tetryl |
479-45-8
|
| Note: Analytes in bold are not identified by EPA Method 8330B.
|
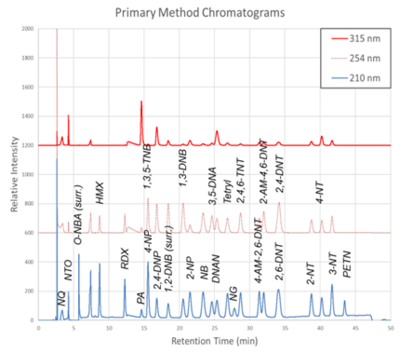
Figure 1. Primary Method labeled chromatograms
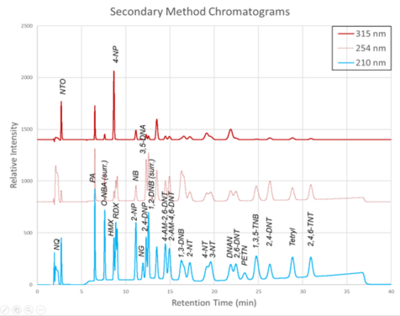
Figure 2. Secondary Method labeled chromatograms
The primary intention of the analytical methods presented here is to support the monitoring of legacy and insensitive munitions contamination on test and training ranges, however legacy and insensitive munitions often accompany each other at demilitarization facilities, manufacturing facilities, and other environmental sites. Energetic materials typically appear on ranges as small, solid particulates and due to their varying functional groups and polarities, can partition in various environmental compartments[3]. To ensure that contaminants are monitored and controlled at these sites and to sustainably manage them a variety of sample matrices (surface or groundwater, process waters, soil, and tissues) must be considered. (Process water refers to water used during industrial manufacturing or processing of legacy and insensitive munitions.) Furthermore, additional analytes must be added to existing methodologies as the usage of IM compounds changes and as new degradation compounds are identified. Of note, relatively new IM formulations containing NTO, DNAN, and NQ are seeing use in IMX-101, IMX-104, Pax-21 and Pax-41 (Table 1)[4][5].
Sampling procedures for legacy and insensitive munitions are identical and utilize multi-increment sampling procedures found in USEPA Method 8330B Appendix A[1]. Sample hold times, subsampling and quality control requirements are also unchanged. The key differences lie in the extraction methods and instrumental methods. Briefly, legacy munitions analysis of low concentration waters uses a single cartridge reverse phase SPE procedure, and acetonitrile (ACN) is used for both extraction and elution for aqueous and solid samples[1][6]. An isocratic separation via reversed-phase C-18 column with 50:50 methanol:water mobile phase or a C-8 column with 15:85 isopropanol:water mobile phase is used to separate legacy munitions[1]. While these procedures are sufficient for analysis of legacy munitions, alternative solvents, additional SPE cartridges, and a gradient elution are all required for the combined analysis of legacy and insensitive munitions.
Previously, analysis of legacy and insensitive munitions required multiple analytical techniques, however the methods presented here combine the two munitions categories resulting in an HPLC-UV method and accompanying extraction methods for a variety of common sample matrices. A secondary HPLC-UV method and a HPLC-MS method were also developed as confirmatory methods. The methods discussed in this article were validated extensively by single-blind round robin testing and subsequent statistical treatment as part of ESTCP ER19-5078. Wherever possible, the quality control criteria in the Department of Defense Quality Systems Manual for Environmental Laboratories were adhered to[7]. Analytes included in the methods presented here are found in Table 1.
The chromatograms produced by the primary and secondary HPLC-UV methods are shown in Figure 1 and Figure 2, respectively. Chromatograms for each detector wavelength used are shown (315, 254, and 210 nm).
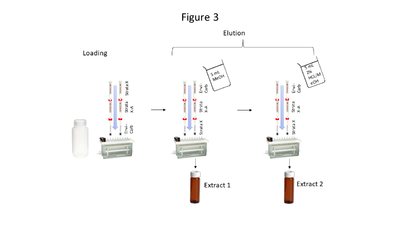
Figure 3. Triple cartridge SPE setup
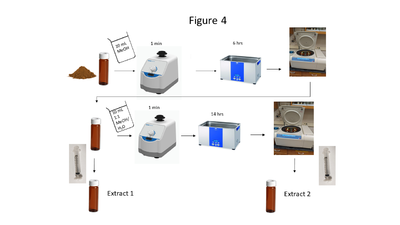
Figure 4. A flow chart of the soil extraction procedure
High Concentration Waters (> 1 ppm)
Aqueous samples suspected to contain the compounds of interest at concentrations detectable without any extraction or pre-concentration are suitable for analysis by direct injection. The method deviates from USEPA Method 8330B by adding a pH adjustment and use of MeOH rather than ACN for dilution[1]. The pH adjustment is needed to ensure method accuracy for ionic compounds (like NTO or PA) in basic samples. A solution of 1% HCl/MeOH is added to both acidify and dilute the samples to a final acid concentration of 0.5% (vol/vol) and a final solvent ratio of 1:1 MeOH/H2O. The direct injection samples are then ready for analysis.
Low Concentration Waters (< 1 ppm)
Aqueous samples suspected to contain the compounds of interest at low concentrations require extraction and pre-concentration using solid phase extraction (SPE). The SPE setup described here uses a triple cartridge setup shown in Figure 3. Briefly, the extraction procedure loads analytes of interest onto the cartridges in this order: StrataTM X, StrataTM X-A, and Envi-CarbTM. Then the cartridge order is reversed, and analytes are eluted via a two-step elution, resulting in 2 extracts (which are combined prior to analysis). Five milliliters of MeOH is used for the first elution, while 5 mL of acidified MeOH (2% HCl) is used for the second elution. The particular SPE cartridges used are noncritical so long as cartridge chemistries are comparable to those above.
Table 2. Primary HPLC-UV mobile phase gradient method concentrations
Method run time = 48 minutes; Column temperature = 25°C
Injection volume = 50 μL; Flow rate = 1.0 mL/min
Detector wavelengths = 210, 254, and 310 nm
|
Time
(min)
|
Reagent Water
(%)
|
MeOH
(%)
|
0.1% TFA/Water
(%)
|
ACN
(%)
|
| 0.00 |
89 |
3 |
3 |
5
|
| 2.00 |
89 |
3 |
3 |
5
|
| 2.20 |
52 |
40 |
3 |
5
|
| 12.5 |
52 |
40 |
3 |
5
|
| 19.0 |
57 |
35 |
3 |
5
|
| 28.0 |
48 |
44 |
3 |
5
|
| 32.0 |
48 |
44 |
3 |
5
|
| 44.0 |
32 |
60 |
3 |
5
|
| 44.1 |
89 |
3 |
3 |
5
|
| 48.0 |
89 |
3 |
3 |
5
|
Soils
Soil collection, storage, drying and grinding procedures are identical to the USEPA Method 8330B procedures[1]; however, the solvent extraction procedure differs in the number of sonication steps, sample mass and solvent used. A flow chart of the soil extraction procedure is shown in Figure 4. Soil masses of approximately 2 g and a sample to solvent ratio of 1:5 (g/mL) are used for soil extraction. The extraction is carried out in a sonication bath chilled below 20 ⁰C and is a two-part extraction, first extracting in MeOH (6 hours) followed by a second sonication in 1:1 MeOH:H2O solution (14 hours). The extracts are centrifuged, and the supernatant is filtered through a 0.45 μm PTFE disk filter.
The solvent volume should generally be 10 mL but if different soil masses are required, solvent volume should be 5 mL/g. The extraction results in 2 separate extracts (MeOH and MeOH:H2O) that are combined prior to analysis.
Tissues
Tissue matrices are extracted by 18-hour sonication using a ratio of 1 gram of wet tissue per 5 mL of MeOH. This extraction is performed in a sonication bath chilled below 20 ⁰C and the supernatant (MeOH) is filtered through a 0.45 μm PTFE disk filter.
Due to the complexity of tissue matrices, an additional tissue cleanup step, adapted from prior research, can be used to reduce interferences[8][2]. The cleanup procedure uses small scale chromatography columns prepared by loading 5 ¾” borosilicate pipettes with 0.2 g activated silica gel (100–200 mesh). The columns are wetted with 1 mL MeOH, which is allowed to fully elute and then discarded prior to loading with 1 mL of extract and collecting in a new amber vial. After the extract is loaded, a 1 mL aliquot of MeOH followed by a 1 mL aliquot of 2% HCL/MeOH is added. This results in a 3 mL silica treated tissue extract. This extract is vortexed and diluted to a final solvent ratio of 1:1 MeOH/H2O before analysis.
HPLC-UV and HPLC-MS Methods
Table 3. Secondary HPLC-UV mobile phase gradient method concentrations
Method run time = 43 minutes; Column temperature = 25°C
Injection volume = 50 μL; Flow rate = 0.8 mL/min
Detector wavelengths = 210, 254, and 310 nm
|
Time
(min)
|
Reagent Water
(%)
|
MeOH
(%)
|
0.1% TFA/Water
(%)
|
ACN
(%)
|
| 0.00 |
75 |
10 |
10 |
5
|
| 2.50 |
75 |
10 |
10 |
5
|
| 2.60 |
39 |
46 |
10 |
5
|
| 9.00 |
39 |
46 |
10 |
5
|
| 9.10 |
33.5 |
51.5 |
10 |
5
|
| 15.00 |
35 |
50 |
10 |
5
|
| 15.10 |
43 |
42 |
10 |
5
|
| 33.00 |
30 |
55 |
10 |
5
|
| 33.10 |
75 |
10 |
10 |
5
|
| 43.00 |
75 |
10 |
10 |
5
|
Table 4. Ionization source and detector parameters
| Parameter
|
Value
|
| Ionization Source |
APCI
|
| Ionization Mode |
Negative
|
| Drying Gas Temperature (°C) |
350
|
| Vaporizer Temperature (°C) |
325
|
| Drying Gas Flow (L/min) |
4.0
|
| Nebulizer Pressure (psig) |
40
|
| Corona Current (μA) |
10
|
| Capillary Potential (V) |
1500
|
| Mass Range |
40 – 400
|
| Fragmentor |
100
|
| Gain |
1
|
| Threshold |
0
|
| Step Size |
0.20
|
| Speed (μ/sec) |
743
|
| Peak Width (min) |
0.06
|
| Cycle Time (sec/cycle) |
0.57
|
The Primary HPLC-UV method uses a Phenomenex Synergi 4 µm Hydro-RP column (80Å, 250 x 4.6 mm), or comparable, and is based on both the HPLC method found in USEPA 8330B and previous work[1][8][2]. This separation relies on a reverse phase column and uses a gradient elution, shown in Table 2. Depending on the analyst’s needs and equipment availability, the method has been proven to work with either 0.1% TFA or 0.25% FA (vol/vol) mobile phase. Addition of a guard column like a Phenomenex SecurityGuard AQ C18 pre-column guard cartridge can be optionally used. These optional changes to the method have no impact on the method’s performance.
The Secondary HPLC-UV method uses a Restek Pinnacle II Biphenyl 5 µm (150 x 4.6 mm) or comparable column and is intended as a confirmatory method. Like the Primary method, this method can use an optional guard column and utilizes a gradient elution, shown in Table 3.
For instruments equipped with a mass spectrometer (MS), a secondary MS method is available and was developed alongside the Primary UV method. The method was designed for use with a single quadrupole MS equipped with an atmospheric pressure chemical ionization (APCI) source, such as an Agilent 6120B. A majority of the analytes shown in Table 1 are amenable to this MS method, however nitroglycerine (which is covered extensively in USEPA method 8332) and 2-,3-, and 4-nitrotoluene compounds aren’t compatible with the MS method. MS method parameters are shown in Table 4.
Summary
The extraction methods and instrumental methods in this article build upon prior munitions analytical methods by adding new compounds, combining legacy and insensitive munitions analysis, and expanding usable sample matrices. These methods have been verified through extensive round robin testing and validation, and while the methods are somewhat challenging, they are crucial when simultaneous analysis of both insensitive and legacy munitions is needed.
References
- ^ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 United States Environmental Protection Agency (USEPA), 2006. EPA Method 8330B (SW-846) Nitroaromatics, Nitramines, and Nitrate Esters by High Performance Liquid Chromatography (HPLC), Revision 2. USEPA Website EPA Method 8330b
- ^ 2.0 2.1 2.2 Crouch, R.A., Smith, J.C., Stromer, B.S., Hubley, C.T., Beal, S., Lotufo, G.R., Butler, A.D., Wynter, M.T., Russell, A.L., Coleman, J.G., Wayne, K.M., Clausen, J.L., Bednar, A.J., 2020. Methods for simultaneous determination of legacy and insensitive munition (IM) constituents in aqueous, soil/sediment, and tissue matrices. Talanta, 217, Article 121008. doi: 10.1016/j.talanta.2020.121008 Open Access Manuscript.pdf
- ^ Walsh, M.R., Temple, T., Bigl, M.F., Tshabalala, S.F., Mai, N. and Ladyman, M., 2017. Investigation of Energetic Particle Distribution from High‐Order Detonations of Munitions. Propellants, Explosives, Pyrotechnics, 42(8), pp. 932-941. doi: 10.1002/prep.201700089
- ^ Mainiero, C. 2015. Picatinny Employees Recognized for Insensitive Munitions. U.S. Army, Picatinny Arsenal Public Affairs. Open Access Press Release
- ^ Frem, D., 2022. A Review on IMX-101 and IMX-104 Melt-Cast Explosives: Insensitive Formulations for the Next-Generation Munition Systems. Propellants, Explosives, Pyrotechnics, 48(1), e202100312. doi: 10.1002/prep.202100312
- ^ United States Environmental Protection Agency (USEPA), 2007. EPA Method 3535A (SW-846) Solid-Phase Extraction (SPE), Revision 1. USEPA Website Method 3535A.pdf
- ^ US Department of Defense and US Department of Energy, 2021. Consolidated Quality Systems Manual (QSM) for Environmental Laboratories, Version 5.4. 387 pages. Free Download QSM Version 5.4.pdf
- ^ 8.0 8.1 Russell, A.L., Seiter, J.M., Coleman, J.G., Winstead, B., Bednar, A.J., 2014. Analysis of munitions constituents in IMX formulations by HPLC and HPLC-MS. Talanta, 128, pp. 524–530. doi: 10.1016/j.talanta.2014.02.013
See Also



